Battery Charger - charging a lead acid battery
A battery can be defined as an electrochemical device that can convert chemical energy within its active materials to electrical energy. If the reaction leading to such a conversion of one form of energy into the other is reversible, then we have a rechargeable or secondary or storage cell. Such cells can be repeatedly recharged after each discharge to reverse the direction of the reaction. For a battery to deliver its intended designed life, it must receive proper charging whenever necessary.
Those cells with irreversible reactions are called primary cells.
A lead-acid battery consists of positive and negative electrodes separated by insulating films called separators. A dilute solution of sulphuric acid is used as the electrolyte. The positive active material is lead dioxide (PbO2) and the negative active material is lead.
Before we delve into the details of Battery Charger, it is necessary to briefly understand a few battery-related matters.
Ampere is the unit for current (which is defined as a continuous flow of electrons). When one coulomb (or one ampere-second) moves past a point in one second, the current is defined as 1 ampere.
Voltage can be taken as the driving force for the electrons to flow in an electronic conductor and the unit is volts. When 1 ampere-second has 1 joule of energy, we say it has 1 volt of electric potential difference.
These two terms can be likened to the overhead water tank in a building. The more the height of the water tank, the higher will be the force with which water will flow. Similarly, the more the diameter of the pipe carrying water from the tank to the user points, the higher will be the volume of water the user receives. The water flowing in the pipe can be compared to the rate at which water flows.
Ampere hour (Ah) is the amount of electricity, and it is a product of current and time.
1 Ah = 1 A *1 hour.
Watts (W) is power, and it is a product of current and volts. The higher units are kW (= 1000 W).
mega watts, MW (=1000 kW) and Giga watts, GW (one billion W (1,000,000,000 Watts).1 W = 1 A * 1 V= VA.
Energy (Wh) is the amount of power supplied in unit time. The higher units are kWh (= 1000 Wh)
megawatt-hour, MWh (= 1000 kWh) and Giga Watt-hours, GWh (=(one billion Wh (1,000,000,000 Watt-hours).
GW units are used to refer to output from large power stations. GWh is used to refer to the production capacity of large electric vehicle (EV) battery industries and large capacity battery storage systems Wh = 1 W* 1 h = 1 Wh
In battery parlance, a battery can be said to possess 1200 Wh (or 1.2 kWh) if its voltage is 12 and its capacity in Ah is 100.
12 V * 100 Ah = 1200 Wh or 1.2 kWh.
The power supplied by unit mass of a battery is termed specific power and the unit is W per kg.
Specific power = W/kg and kW/kg.
Similarly, the energy supplied by unit mass of a battery is termed specific energy and the unit is Wh per kg.
Specific energy = Wh / kg and kWh / kg. (Also written as Wh kg-1)
Similarly, the power supplied by unit volume of a battery is termed power density and the unit is W per litre.
Power density = W / litre and kW / litre.
The energy supplied by unit volume of a battery is termed energy density and the unit is Wh per litre.
1 W = 1 J per second
Energy density = Wh / litre and kWh / litre. (Also written as W L-1 or W l-1)
The discharge-charge reaction of a lead-acid cell is
Pb (NP) + PbO2 (PP) + 2H2SO4 Discharge ⇔ Charge PbSO4 (PP) + PbSO4 (NP) + 2H2O (Near PP)
Note: NP = negative plate= anode during discharge=donor of electrons during discharge. PP = positive plate = cathode during discharge = acceptor of electrons during discharge
The roles of the electrodes will be reversed during a charge; anode will behave as cathode and vice versa. The electron acceptor will now release electrons and the donor will receive them.
The term thermodynamic free energy is a measure of the work that can be extracted from a system. In the case of galvanic cell, the electrical work is done through the movement of charged particles due to the chemical interaction between the reactants to produce the resultants (products).
Hence, the energy is given in terms of ΔG, the change in the Gibb’s free energy, which represents the maximum amount of chemical energy that may be extracted from the energy conversion processes.
If E is the emf (electromotive force or voltage or potential) of the cell and the process, which is taking place (i.e., a discharge of a lead-acid cell), is associated with a passage of n Faradays (F) per mole of reactants from one electrode to the other, then the electrical work done by the cell is given as nFE. The corresponding increase in free energy is equal to the electrical work done on the system. Hence,
ΔG = nFE or
ΔG = -nFE or
-ΔG° = nFE°
(under standard conditions; E° refers to standard electrode potential or standard cell voltage).
Gibbs equation
(What is meant by standard conditions?: 25°C or Celsius (298.1°K or Kelvin), 1 bar pressure, and the activity (which can be approximately taken as the value of concentration) of the reacting species, Pb2+, is one).
This equation is called the Gibbs equation.
Gibbs equation connects the cell voltage to the change in free energy (DG). If the reaction occurs spontaneously (e.g. a discharge of a lead-acid cell), ΔG is negative (energy is liberated) and emf is positive i.e., a charge of nF will flow spontaneously in the direction as assumed in the cell reaction.
On the other hand, if ΔG is positive, it enables the system to perform the phenomenon of electrolysis (i.e., during a charge of lead-acid cell).
EMF of a cell
The emf of the cell is an intensive thermodynamic property i.e., independent of both the mass of the reactants and the size of the cell. The intensive property (as opposed to the extensive property) does not depend on the mass of the reactants and hence the size of the battery. Whether you have a few milligrams or a few kilograms of the materials, the system will show the same voltage and it cannot be increased by increasing the mass of the material. The individual electrode potential is an inherent electrochemical property of that electrode material, and one cannot change its value under similar conditions.
Examples for intensive property are the voltage of the electrodes and cells; on the other hand, the extensive property depends on the amount of substance, for example, mass, volume, energy, ampere hour and watt hour. Thus, 4.5 grammes of lead dioxide active material in a lead-acid cell will deliver one ampere hour (Ah) theoretically, but if you have 45 grammes, it will deliver ten times the Ah. So, it is an extensive property; bur in both cases the electrode potential will be the same, namely, 1.69 V. Similar arguments can be put forward for lead and sulphuric acid active materials.
The standard cell potential (E°) is related to the standard free energy change (DG°) as given above.
The emf of a lead-acid cell can be determined from the expression
ΣΔGº ƒ of products – ΣΔGº ƒ of reactants
Where ΔG°ƒ refers to Standard free energy of formation of the reacting species.
Standard free energy of formation
Table 1
Standard free energy of formation, ΔG°ƒ of chemical species participating in the cell reaction
(Hans Bode, Lead-Acid Batteries, John Wiley, New York, 1977, Appendix IV, p. 366.)
| Reactants/Products | Numerical Value (k cal mole−1 ) |
|---|---|
| PbO2 | -52.34 |
| Pb | 0 |
| H2SO4 | -177.34 |
| PbSO4 | -193.8 |
| H2O | -56.69 |
The overall reaction is written as
Pb + PbO2 + 2H2SO4 ⇄ 2PbSO4 + 2H2O E° = 2.04 V.
ΔG° = ΣΔGº ƒ of products – ΣΔGº ƒ of reactants
By substituting the respective values of (which we get from Standard Textbooks, for example, [1. Hans Bode, Lead-Acid Batteries, John Wiley, New York, 1977, Appendix IV, p. 366]
= [2(−193.89) + 2(−56.69)] − [0 − (−52.34) + 2(−177.34)]
= −94.14 kcal mole−1
= −94.14 kcal mole−1 × 4.184 kJ mole−1 (to convert kcal to kJ multiply by 4.184 )
= −393.88 kJ per mole
E° = -ΔG°/nF
= −(−393.88 × 1000)/2 × 96485
= 2.04 V for a lead-acid cell
The standard cell voltage of a lead-acid cell is 2.04 V
and the overall or the total cell reaction of a lead-acid cell is written as:
Pb + PbO2 + 2H2SO4 Discharge⇔Charge PbSO4 (PP) + PbSO4 (NP) + 2H2O (Near PP)
Before we go into the details of charging and discharging of a lead-acid cell, we should have some knowledge of certain terms used in Electrochemistry.
We already know the meaning of standard conditions.
When we disturb the cell reaction (whether in the forward direction or reverse direction), we say the cell is in a disturbed condition and not in equilibrium conditions.
Whenever an electrochemical system is disturbed, there will always be difference from the standard potential. Thus, if a lead-acid cell is forced in the discharge direction the cell voltage decreases by a certain value, which depends on the magnitude of the current. The higher the current value, the more will be the deviation from the standard value.
Now the cell voltage will be
EDisch = E° – δV.
The value of EDisch will be lower than that of E°.
On the contrary, if the cell is forced in the reverse direction (i.e., charging mode), the cell voltage will increase by a certain value which again depends on the magnitude of the current.
ECh = E° + δV.
The value of δV is called overvoltage or overpotential and is denoted by the symbol η.
The value of δV will be negative for a discharge reaction and positive for a charge reaction.
This phenomenon of decease or increase in voltage of the cell is called polarization and the electrodes are said to be in a polarized state.
So, we rewrite the equations as follows:
EDisch = E° – η.
ECh = E° + η.
Thus it is seen that during a discharge
EDisch < E° and
During a charge
ECh > E°.
What are the reasons for this deviation of voltage?
There are a few causes for this deviation:
- Loss due to internal resistances (IR) (ηohmic)
- Activation polarization due to charge transfer on the two electrodes during the start of the process ηt.
- Concentration polarization due to the depletion of electrolyte and other participating species (ηc).
The losses due to IR polarization can be mitigated by using electrode current collectors and electrolyte having better conductivities. A separator with a lower resistance will also help.
Activation polarization is related to the transfer of charge carriers across the phase boundaries of the electrode and this process is designated as transfer reaction. The transfer overvoltage due to charge transfer reactions on the two electrodes can be very much reduced in battery electrodes by having a compatible porous structure. The latter increases the actual internal surface area (BET surface area, which includes the areas of pores, cracks, and fissures) as opposed to the apparent surface area obtained by the multiplication of the dimensions, length, and breadth) available for the reactions.
Current density
This in turn reduces the current density (that is, ampere per square cm). Thus, a plate with an aggregate porosity of 40 % will lead to higher losses due to activation polarization than one with 50 % porosity.
Concentration polarization (ηc) will be more if the reaction products (lead sulphate and water molecules, in the case of a lead-acid cell) are not transferred away from the electrode surfaces to make way for fresh reactants (e.g. lead ions from both the electrodes and sulphate ions from the electrolyte in the case of a lead-acid cell). ηc will be more pronounced towards the end of a discharge reaction. Inside a cell, the transport of ions is done by diffusion and by migration.
Diffusion is caused by the differences in in concentration, whereas migration is caused by forces of the electric field.
Diffusion can occur in the bulk of the electrolyte or the separator: as the ions are generated at one electrode and are consumed at the other electrode, the ions have to move between the electrodes.
It also occurs in the porous electrodes as the electrochemical reaction proceeds. The reaction products can move within the active mass to their final location by diffusion.
The share of the total current that is carried by the particular ionic species (charged particles) by migration is a function of their transference number. In a binary electrolyte, dissociated into cations and anions the transference numbers are related by the equation
ɩC + ɩA= 1,
where ɩC + ɩA denote the transport number of cations and anions.
Transference numbers depend on concentration of the ions and on temperature. In binary salt solutions they are nearly close to 0.5. Thus both ionic species share equally in ionic conductivity.
Significant deviations occur in strong acids and alkalis due to the higher ionic mobility of protons (H+) and hydroxyl ions (OH–). The values for the battery electrolyte sulphuric acid (dissociated into H+ and HSO2-4) and potassium hydroxide (dissociated into K+ and OH–) are given below. 4
ιH+ = 0.9; ɩHSO42- = 0.1; ιK+ = 0.22; ιOH-= 0.78
The transference number is a measure of how much the concentration of the particular ion is influenced by migration due to the current flow. A smaller value is indicative of smaller influence on the migration processes and a higher value indicates greater influence on the migration process.
2. D. Berndt, in Battery Technology Handbook, Ed. H.A. Kiehne, Second Edition, 2003, Marcel Dekker, Inc., New York, Table 1.2.
3. J S Newman. Electrochemical Systems. Englewood Cliffs: Prentice-Hall, 1991, p 255.
4. S U Falk, A J Salkind. Alkaline Storage Batteries. New York: John Wiley & Sons, 1969, p 598
To make it clear, we must understand how the discharge reaction is proceeding. As soon as the battery terminals are connected to the consuming apparatus, electrons begin to flow from the negative plate to the positive terminal through the external circuit. Inside the cell, it is the duty of the charged particles to take care of the current flow. The charged particles are protons (H+) and bisulphate ions (HSO¯4 ).
During a discharge, negative HSO¯4 ions (in this case, bisulphate ions from the electrolyte sulphuric acid which dissociates as H+ and HSO¯4 ) move towards the negative plate. These negative ions are combined with the active material, Pb, producing, lead sulphate, PbSO4. The reaction also produces a positively charged hydrogen ion called proton) which migrates away. The two electrons released as a result of the anodic reaction of lead active material, reach the positive terminal via the external circuit.
Negative plate or negative half cell reaction: Pb + HSO¯4 ⇄ Pb2+ + SO42- +H+ + 2e– E°= -0.35 V
The bivalent lead ions and sulphate ions immediately combine to form lead sulphate and get deposited on the negative plate as lead sulphate.
So far, we have seen the picture of the negative plate reactions.
Now let us see what happens simultaneously on the positive plate.
The electrons from the negative plate, after reaching the positive terminal, react with the positive active martial, PbO2, to form lead sulphate and two water molecules.
Positive plate or positive half cell reaction: PbO2 + 3H+ + HSO¯4 + 2e– ⇄ Pb2+ + SO4 2- + 2H2O E° = 1.69 V
The bivalent lead ions (Pb2+) and sulphate ions ( ) immediately combine to form lead sulphate and get deposited on the positive plate as lead sulphate.
Dissolution-deposition or dissolution-precipitation mechanism
This type of reaction, where the lead and lead dioxide dissolve as lead ions and get immediately deposited as lead sulphate on the respective electrodes is happening through a dissolution-deposition or dissolution-precipitation mechanism.
Now by combining the two half-cell reactions, we have
Negative plate or negative half-cell reaction: Pb + HSO¯4 ⇄ Pb2+ + SO42- +H+ + 2e–
Positive plate or positive half-cell reaction: PbO2 + 3H+ + HSO¯4 + 2e– ⇄ Pb2+ + SO42- + 2H2O
Overall or the total reaction: Pb + PbO2 + 2H2SO4 Discharge⇔Charge 2PbSO4 + 2H2O
This reaction theory was proposed by Gladstone and Tribe in 1881, but the lead-acid cell was invented in 1859 by Raymond Gaston Planté, French Physicist.
J.H. Gladstone and A. Tribe, Chemistry of the Planté and Fauré Accumulators, Nature, 25 (1881) 221 & 461.
J.H. Gladstone and A. Tribe, Chemistry of the Planté and Fauré Accumulators, Nature, 26 (1882) 251, 342 & 602; 27 (1883) 583
The discharge reaction will proceed until about half of the active materials is converted to lead sulphate for a slow rate of discharge, such as 20- or 10-hour rates. By this time, the resistivity of the active materials would have increased to such a value that further discharge would result in very fast drop in the cell voltage. Normally, the cell voltage is not allowed to go to lesser than 1.75 V per cell.
Deep discharges beyond 80 % depth of discharge (DOD) will make the subsequent recharge more difficult.
As soon the lead dissolves as lead ions during the discharge reaction, it combines with the sulphate ions and gets deposited on the negative plate. The lead ions or lead sulphate molecule do not go far away from the negative plate. This is because the solubility of lead sulphate in dilute sulphuric acid solutions is very low. It is of the order of above 1 mg per litre, the deposition of bivalent lead ions to lead sulphate will be faster at places where there are high concentrations of electrolyte. As the discharge proceeds further, the solubility of lead sulphate in the electrolyte increases up to 4 mg per litre.
This is so because the acid becomes more dilute because of further discharge and in such dilute acids, the solubility of lead sulphate is higher, up to 4 mg per litre..
The lead sulphate so deposited will continue to grow to various sizes of crystals both on the surface and cracks and crevices. . The film will be discontinuous in structure. During a slow discharge process, this discontinuous form of lead sulfate structure helps the inner portions of active materials to participate in the reaction as it provides an open structure that facilitates easy entry of ions. Therefore, the discharge process can proceed deep into the interior of the plate.
On the contrary, at high rates of discharges, the surface is blocked by the discharge product, PbSO4, which forms a continuous structure without any break. Thus, further reactions in the interiors of the plates are hindered and that is why we cannot get expected capacity at higher discharge rates.
Charging of lead-acid batteries
During a charging reaction, the reverse phenomena occur, the current flow is reversed and oxidation takes
place at the positive electrode and reduction at the negative electrode.
Table 2
Characteristics of the two electrodes during charge and discharge
| Electrode | Discharging | Charging |
|---|---|---|
| Negative Plate |
Porous (Spongy) Lead Anode Gives up 2 electrons Pb -2e- → Pb2+ Voltage decreases (becomes less positive). Converted to PbSO4 |
~ 40 % Pb + ~60% PbSO4 Cathode Absorbs 2 electrons Pb2+ in PbSO4 takes up 2 electrons Voltage decreases (becomes more negative) Recoverted to Pb metal H2 evolved during overcharge |
| Positive Plate |
Porous Lead dioxide Cathode Absorbs 2 electrons Pb4+ (from PbO2) + 2e- → Pb2+ Voltage decreases (becomes less positive). Converted to PbSO4 |
~ 50 % PbO2 + ~50% PbSO4 Anode Releases 2 electrons Pb2+ in PbSO4 becomes PbO2 Reconverted to PbO2 Voltage increases O2 evolved during overcharge |
Figure 1
Change values of potential for lead-acid cell during charge and discharge reactions
The cell voltage is a combination of two values at any stage of the functioning of a galvanic cell
Thus
Cell voltage = Positive electrode potential – Negative electrode potential
Therefore
Lead-acid cell’s Open circuit voltage or Equilibrium voltage = 1.69 – (-0.35) = 2.04 V
At or near the end of a discharge, the cell voltage, EDisch = 1.50 – (- 0.20) = 1.70 V
At or near the end of a charge, the cell voltage, ECh = 2.05 – (-0.65) = 2.70 V

Battery charger - Charging coefficient
Rechargeable batteries require to be charged to get back the Ah capacity spent in the previous discharge.
The amount of Ah required to bring the battery to the previous fully charged state in comparison with the earlier output will be 10 to 15 % more. This ratio of charge input to the previous output is called the charge coefficient
Charge coefficient = Input Ah / Previous output Ah = ~ 1.1 to 1.2.
That is, about 10 to 20 % extra Ah should be put in to compensate for the secondary reactions, which are constituted by the water-splitting overcharge reactions and grid corrosion reactions. Also, a small part will be lost due to internal resistance.
Battery charger - Charging Efficiency of lead acid battery
Ampere hour efficiency
(Ampere hour or coulombic efficiency and energy or watt-hour efficiency)
From the foregoing arguments, it can be seen that we have to define what is termed “charging efficiency”.
Ampere hour efficiency
Indian Standard IS 1651 describes the test procedure as follows:
- A fully charged battery shall be subjected to a discharge at ten hour rate to an end voltage of 1.85 volts per cell.
- The exact Ah output shall be calculated.
- The battery is now recharged with the same number of ampere-hours at the same current.
- The battery is now subjected to a second discharge as before.
- The Ah (Coulombic) efficiency= ηAh = Ah delivered during second discharge / The Ah input.
Energy or watt-hour efficiency
The watt-hour efficiency shall be calculated by multiplying the ampere-hour efficiency obtained as described above by the ratio of average discharge and recharge voltage.
Energy or watt hour efficiency = ηWh = ηAh * (Mean discharge voltage / Mean charge voltage)
The ampere hour (or coulombic) efficiency of charging of the lead-acid cell in the case of an input equal to 100% of the previous discharge at the same rate is nearly equal to 95% and the energy or watt hour efficiency is about 85-90%. Indian Standards (IS 1651) also specifies a minimum ampere-hour efficiency of 90 % and a minimum watt-hour efficiency of 75%.
The charging efficiency is limited by the positive plate rather than the negative one. When about three fourth of the lead sulfate on the positive electrode has been converted back to lead dioxide and the water cannot diffuse sufficiently fast into the inner plate porous structure, secondary reactions like the evolution of oxygen take place. For some period of time, the charging current is distributed between the primary process of converting PbSO4 to PbO2 and the secondary overcharge reactions. If charging continues for a sufficiently long time so that almost all the lead sulphate would have been converted to lead dioxide all of the charging current goes for the secondary reactions.
Charging Voltage of a battery charger
As explained earlier
ECh > E°.
So, we have to supply a little higher voltage in order to facilitate this reaction. Normally, a good charger will be designed with sufficiently higher voltage source for charging. It is a good rule of thumb that for a 2 V cell one must provide at least 3 V so that the cell can attain full charge by reaching a voltage of 2.7 V per cell. But we must take into consideration the losses in the cable, etc.
Hence for a 12 V battery, the battery charger should provide at least 18 to 20 V.
If this voltage is reduced to lower than 15 V. then the battery cannot attain the fully charged state.
During a recharge: 2PbSO4 + 2H2O → PbO2 + Pb + 2H2SO4
The lead sulfate on both electrodes dissolves as lead ions and gets immediately deposited as lead on the negative plate and as PbO2 on the positive electrode.
At the positive plate
PbSO4 + 2H2O → PbO2 + 4H+ +SO4 ²- + 2e–
The electrons travel to the negative plate for further reaction
At the negative plate
PbSO4 + 2e– → Pb +SO4 ²-
Since sulfate ions are reproduced on both plates, they combine with the protons to form sulphuric acid and so the specific gravity of the electrolyte increases.
Battery Gassing
Until now we have seen only the useful reactions during the charging process. But there are some side reactions or secondary reactions occurring in the overcharge periods. The two main secondary or side reactions are:
- Electrolysis of water and
- Corrosion of positive grids
These reactions can be represented as follow:
Water electrolysis
2H2O → O2 ↑ + 2H2 ↑ (On both plates of excess flooded, electrolyte lead-acid cells)
Oxygen from the positive plate and hydrogen from the negative plates evolve and are vented out to the atmosphere through the vent plug holes.
But in a valve regulated lead acid battery (VRLA) cell, oxygen does evolve, but not hydrogen. The oxygen so evolved also is not allowed vent out but diffuses via the voids available in the absorptive glass mat (AGM) separator and reacts with the negative active material to regenerate water molecules. This is the step that makes it possible for the VRLA cell to thrive without topping up with water.
2H2O → O2 + 4H+ + 4e – On the positive plate of Starved electrolyte or VRLA cells
Corrosion of positive grids in a lead acid battery
In both types of lead-acid cells positive grid corrosion occurs in the same way:
Grid corrosion: Pb + 2H2O → PbO2 + 4H+ + 4e–
If a platinized platinum electrode is made a cathode, hydrogen is evolved almost at the reversible
hydrogen potential of the solution. With other electrodes, e.g. lead, a more negative potential is required
for this reaction to occur.
Until the cell voltage reaches a value of 2.3 V, there is negligible gassing. But the gassing begins at 2.4 V per cell. Beyond 2.4 V, the gassing is more and hence the charging efficiency will be reduced. At 2.5 V, the gassing will be copious, and the temperature of the battery electrolyte will begin to rise. Now there is sufficient gassing to provide agitation of the electrolyte and the specific gravity begins to equalize. When the battery is idle, the specific gravity of the electrolyte will be a little higher at the bottom than at the top level. This is aggravated if the cells are taller.
The lead-acid battery can be charged at any rate that does not lead to excessive gassing, high temperature and very high voltage at the terminals. A fully discharged battery can absorb a high rate of charge at the beginning of charging without gassing and any appreciable rise in voltage and temperature.
At some time of the charging process, when almost all the lead sulfate has been converted to lead dioxide in the positive plat, the secondary reactions take predominance. These are the water electrolysis reaction and positive grid corrosion, as given earlier.
Such positive grid corrosion starts right from the formation stage (or in the case of jar formation) from th first charge. This corrosion is the most inimical aspect to the life of the lead-acid battery. Since the corrosion of the positive grid is occurring whenever the cell enters over charge region, a part of the grid structure gets converted to lead dioxide and so the weight of the grid comes down a little at each corrosion period. Ultimately, a stage will be reached when electrons from the reaction sites on the grids cannot travel to the bus bar, because of the no-availability of a continuous grid structure
As a consequence, a part of the active material cannot participate in the energy production process and the capacity declines, leading to the end of life of the battery.
The manufacturers of lead-acid cells try to mitigate this problem by the inclusion of alloying elements which increase the corrosion resistance of the lead alloys. Some such alloying constituents are arsenic (As) and silver (Ag) in fractional percentages. As a rule, the amount of As will be around 0.2 % and Ag about 0.03 to 0.05 % in the positive alloys.
Battery charger - current acceptance meaning
The current acceptance is dictated by the design of the cell. For example, a similar Ah battery assembled with a greater number of plates (that is the plates will be thinner), can accept a higher charging current because of the enhanced surface area. For detailed procedures for measuring the charge efficiencies of the individual plates, the readers are referred to an article by K. Peters. [8]
The charge-acceptance of the negative plate is greater than that of the positive plate (See Figure 1) which is mainly due to its coarser, more open and pore structure that easily admits acid diffusion into the plate interior. The positive begins to get overcharged at 70–80% SOC, depending upon several design factors. Some inner parametric design factors are pore structure, true surface area, etc. Other external parameters are the charging current in amperes, temperature of the electrolyte, etc.
The charge-acceptance of the negative plate is higher and it goes into overcharge region at comparatively later period, 90% SOC [8. K. Peters, A.I. Harrison, W.H. Durant, Power Sources 2. Research and Development in Non-mechanical Electrochemical Power Sources, Pergamon Press, New York, USA, 1970, pp. 1–16.]
[9. A.M. Hardman, Journal of Power Sources Vol. 23, Year 1988, page, 128].

At some point, however, secondary reactions begin at the negative electrode, primarily the reduction of hydrogen ion (proton) to hydrogen gas by simple electron transfer (occurring at potentials far lower than -350 mV which is the negative plate reversible potential, E° value.), at about -0.6 to 0.95 V:
2H+ + 2e− → H2 ↑
One such important impurity accumulating on the negative plate is antimony (Sb), which is deposited due to the phenomenon called antimony-migration in cells containing a relatively higher amount of antimony in the grids. Although antimony is an essential component of the grid alloy for most of the lead-acid cells, it has a negative impact on the cell’s performance.
During the corrosion stage of the charging (towards the end of charge of each cycle), the positive grid comes under anodic attack and antimony passes into solution as Sb5+ ions, a portion of which is absorbed by the positive active material where it promotes self-discharge due to local cell formation. The remainder of the antimony so dissolved gets deposited as Sb3+ on the cathode surface (the negative plate surface) (“antimony migration”) and due to its lower hydrogen over potential than lead it causes premature evolution of hydrogen. Later, during copious gas evolution periods the antimony may, under favourable conditions, be released to some extent as stibine gas (SbH3), when it combines with protons.
Under favourable conditions, a similar reaction with arsenic (As) may also occur releasing arsine (AsH3), which is a poisonous gas. Therefore, this alloying constituent is naturally avoided where cells are used in closed surroundings, such as a submarine.
Thermodynamically, this occurs at a lower potential than the primary charging reaction but, as with oxygen generation at the positive electrode, the overpotential for hydrogen generation on the lead electrode is relatively great (about -0.650 V) and so recharge can be largely completed before hydrogen evolution begins in full.
These gases are vented from the cell through the vent plug holes. Both the plates are affected by impurity effects on overpotential, and thus perfectly efficient recharge of both plates is not possible. For example, if you combine the oxygen evolution reaction potential with that of hydrogen evolution, we have
1.95 + (-0.95) = 2.9 V for copious gas evolution.
Another point to be noted is that as per the fundamental laws, water should decompose at 1.23 V and oxygen should evolve on a positive electrode at this potential. But this is not the case in a practical cell. If it happens, the stability of the lead-acid cell itself will be a question. The standard positive plate potential (E° = 1.69 V) is about 0.46V above the voltage at which water should decompose (1.23V). The reason is again overvoltage. That is, the voltage for oxygen evolution on lead dioxide in sulphuric acid solution lies far above the E° value of positive plate at 1.95V.
Thus the oxygen evolution reaction on lead dioxide in sulphuric acid solution is inhibited, being 0.26 V (1.95-1.69 = 0.26) above E° value of positive plate and about 0.72 V above the water decomposition potential (1.95-1.23 = 0.72V) and hence oxygen does not evolve until the overvoltage value is reached in a strictly pure solution.
Similarly, the evolution of hydrogen on lead in sulphuric acid solution is strongly inhibited because of the hydrogen overpotential on lead. This overpotential value is about 0.6 V more negative and is below the standards electrode potential of lead in sulphuric acid solution, E° = -0.35V. Hence hydrogen evolution reaction will not hinder the full charge of negative plate until the electrode value attains -0.95V in a strictly pure solution. This is the reason why the negative plate has a better charge efficiency than the positive plate.
But, in a practical cell, this stage is reached far before this voltage. In fact, this 2.9 V is not at all realized in a practical cells, because the reactions due to impurities take predominance and so the full gas evolution by volume (H2 : O2 = 2:1) is attained at about 2.6 V. However, if the impressed charging voltage is excessively high, then this value of 2.9 V may be reached, particularly, Sb-free alloy batteries may attain a value of 2.8 V and with antimonial cells the value will be lower by 0,2 V, say 2.6 V.
As the cycling proceed further, the gassing value will be very much reduced in the case of antimonial cells, while the other cell is almost free from this effect. This drastic reduction is due to the phenomenon called “antimony migration” as explained earlier.
Naturally, the voltage difference of the new and the cycled batteries increases from 250 mV to 400 mV. This will result in the active materials’ inability to accept charge and almost all the current generates hydrogen and oxygen. Figure 3 illustrates this aspect [10. Hans Tuphorn, Chapter 17, Figure 17.2 in Battery Technology Handbook, Ed. H.A. Kiehne, Second Edition, 2003, Marcel Dekker, Inc., New York.]

How does a 12v battery charger work?
For charging a battery, the positive output lead is connected to the positive terminal of the battery and so the negative to the negative terminal. The charger is then connected to the AC mains supply through a suitable way.
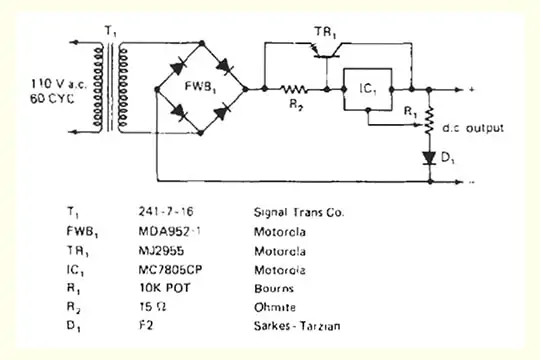
The AC input is converted to DC by a rectifier circuit which has a step-down transformer to convert to the required voltage. A rectifier converts the bi-directional alternating flow of current (AC) to unidirectional flow. Thus, it maintains a constant polarity across the load. A bridge rectifier configuration is used to rectify the stepped down low voltage AC into DC and is further smoothed by a high value electrolytic capacitor (filtering circuit).
This filtered DC is fed to an electronic circuit which regulates the voltage into a constant level and is applied to the battery requiring a charge,
The charger has indicators for the current (ammeter), voltage (voltmeter), and also in special cases a timer and an ampere-hour meter.
The battery is charged as per the manufacturer’s instructions
Battery charging procedure - Battery Charger
The battery requiring to be charged shall be cleaned outside thoroughly and the terminals, after removing the corrosion product, if any, shall be given a thin coating of white Vaseline. The electrolyte level will also be checked. Topping up need not be done at this time unless the level is below the height of the separators.
The charger intended for charging the battery shall have adequate specifications, like voltage and current out puts. For example, a 12 V battery needs an output C voltage of at least 18 V. The current required depends on the capacity of the battery and on the time within which the battery is required to be charges. Normally, a battery will be charged at 0ne tenth amperes of the Ah capacity of the battery. Thus, a 100 ah battery will require at least 10 amperes output for normal charging. If it is to be charged quickly, 15 amperes output will be required.
An input of about 110 % of the capacity is required for a fully discharged battery. But, if the battery is already partially charged, we should know the SOC. Whatever it is, the voltage and specific gravity are the two important parameters to be monitored to determine the state of charge. The specific gravity value should be read from the label on the battery. A fully charged battery will normally reach 16.5 V and more, if it is in good condition. If it is an aged battery, this voltage cannot be easily reached.
This is predominantly because of the secondary reactions like gas evolution due to the electrolysis of the water in the electrolyte and heating effects due to already built-up resistances due to accumulated lead sulfate.
The battery is placed on an insulating material like rubber sheet or a wooden bench. The charger lead should have adequate current-carrying capacity. Normally, 1mm square copper wire can carry 3 amperes direct current (DC) safely. After ensuring that the charger is in the off position, the charger leads will be connected to the respective terminals, i.e., positive to positive and negative to negative. The voltage, the specific gravity and temperature readings will be recorded in a log sheet, a model of which is given below:
Battery charging record template

Readings shall be recorded every hour.
The cadmium readings will indicate whether a particular plate has attained full charge or not. The cadmium reference electrode is an insulated cadmium rod with a copper wire soldered to the top end. The bottom end will be immersed in the electrolyte, so that it just touches the liquid, and it should not come into contact with the plates or other lead parts inside.
For a fully charged positive plate, the cadmium reading will be 2.4 V and more and for negative plate, minus 0.2 V and less.
Table 4
Reactions in lead-acid cell and the corresponding cadmium potential readings
Cadmium potential readings
| Reactions | Potential Values | Cadmium Readings |
|---|---|---|
| Oxygen evolution potential 2 H2O → O2 + 4 H+ + 4e- | 1.95 to 2.00 V | 2.00 - (-0.4) = 2.4 V |
| Standard Electrode Potential of Positive plate PbO2/PbSO4/H2SO4 | 1.69 V | [1.69 – (-0.4) = 2.09 V] |
| End of discharge of positive plate | 1.40 to 1.5 V |
1.40 – (-0.4) = 1.8 V 1.50 – (-0.4) = 1.9 V |
| Standard Hydrogen Electrode Potential (SHE) 2H+ + 2e- → H2 | 0.00 V | 0.00 V |
| End of discharge of negative plate | -0.15, -0.20, -0.25 V (For different current densities) | -0.15 – (-0.4) = 0.25 V -0.20 – (-0.4) = 0.20 V -0.25 – (-0.4) = 0.15 V |
| Standard Electrode Potential of Negative plate Pb/PbSO4/H2SO4 | -0.35 V | [-0.35 – (-0.4) = 0.05 V] |
| Cadmium reference electrode E° value Cd/Cd2+ | -0.40 V | -0.40 V |
| Hydrogen evolution potential- 2H+ + 2e− →H2 (For a commercial cell) | -0.60 V | -0.60 – (-0.4) = -0.20 |
| Hydrogen evolution potential 2H+ + 2e− →H2 For a pure experimental cell | -0.95 V | -0.95 – (-0.4) = -0.55 |
Working principle of a battery charger
At the end of charging, a 12 V battery may attain a terminal voltage of 16.5 and above. After maintaining the terminal voltage at this level for an hour, the charging can be terminated. When the battery is nearing 16. 0 V, approved water can be added, if needed.
Near the end of charging, heavy gassing will be observed from the battery. No open flames should be brought near the charging room. The gases evolve in the ratio of their combination, that is hydrogen 2 parts and oxygen 1 part. Hence if these gases are allowed to accumulate in the charging area without proper ventilation, it is likely that a spark or an open flame will ignite the gases and they will combine with explosive violence, damaging the battery and its surroundings and also hurting the nearby people.
The lower limit for explosive mixture of hydrogen in air is 4.1%, but, for safety reason hydrogen should not exceed 2% by volume. The upper limit is 74%. Heavy explosion occurs with violence when the mixture contains stoichiometric ratio of these gases (2 parts of hydrogen to 1 of oxygen). This condition is obtained inside an overcharging battery with vent plugs tightly screwed to the cover. Hence it is advised to keep the vent plugs loosely over the vent holes and not to screw down tightly.
Different Methods of charging batteries and Different types of battery chargers
Although there are various methods of charging lead-acid cells, all of them have one common aim of converting the reaction products, namely lead sulfate on both the plates to the respective active materials, PbO2 on the positive electrode and Pb on the negative electrode.
2 PbSO4 + 2 H2O → PbO2 + Pb + 2 H2SO4
There are a number of variants in charging regimes. But in all these methods, only two basic principles are used: constant current and constant voltage charging methods. The several methods available combine these two principles to achieve their aims.
Selection of the appropriate method of charging depends upon the type, design and service conditions and the time available for charging. All these charging methods use many methods to control and finish the charging process.
These methods may be classified into the following:
Table 5
Classification of the methods of different battery charger & battery charging methods
Different battery charging methods
| Constant-current based methods (CC) | Constant-voltage based methods (CV or CP) | Combination methods | Taper charging | Special methods |
|---|---|---|---|---|
| Single-step CC charging method | Constant Voltage method | CC-CV method | Single-step taper charging method |
1. Initial charge 2. Equalization charge 3. Opportunity charging 4. Gas-controlled charging 5. Trickle charging 6. Boost charging 7. Pulse charging 8. Fast or Rapid charging |
| Two-step CC charging method | Current-limited or modified CV method | Two-step taper charging method |
Single step Constant-current based charging method (CC method) Battery charger
When recharge is required to be finished in a short and when the user wants to know the input in terms of Ah, the constant-current charging method may be employed. Constant-current charging is preferred when the when the previous output is known, so that 5-10% overcharge can be effective to bring the battery back to 100% SOC. This will also ensure that the correct input is given so that the life of the battery is not adversely affected by undue overcharge. A normal recharge time for this method is 15 to 20 hours.
In this method, the current is maintained constant throughout the charging period.
A charge current of 5 to 10 % of the 20-hour capacity is recommended.
To compensate for the increase in the back emf of the battery while charging, the charging current has to be maintained constant either by varying the series resistance used or by increasing the transformer voltage. Usually, the series resistance is varied to keep the current constant.
This method is the simplest and less expensive method of charging. But it has the disadvantage of lower charge efficiency. This is due to some power being dissipated in the resistance and also partly due to the current used for splitting the water once the battery reaches 2.5 V per cell. The battery starts gassing as the battery is charged to about 70 to 75% charge. This method of charging always results in slight over charging and vigorous gassing especially at the end of charging.
A generalized picture for the constant-current charging method is given Figure 5. The charging characteristics are given in Figure 6


Two-step constant-current charging method Battery charger
Two charging rates, starting rate and finishing rate, are used in the two-step constant-current charging method. The finishing rate is normally half of the starting rate. The finishing rate is commenced when the battery begins to evolve gases. This is generally a preferred method employed for the bench charging of batteries. The charging characteristic may be seen in Figure 7 [11. P G Balakrishnan, Lead Storage Batteries, Scitech Publications (India) Pvt. Ltd., Chennai, 2011, page 12.8].

Constant voltage or potential charging methods Battery charger
Constant voltage or potential (CV or CP) charging method employs a source voltage that is maintained at a constant level throughout the charging period. Usually, this voltage will be between 2.25 and 2.4 V per cell.
This method is the recommended method for charging valve-regulated lead-acid (VRLA) cells and batteries. One need not worry about the depth of discharge (DOD) of the previous discharge when charging a VRLA battery by CV method. The VRLA Batteries can be charged without any detrimental effects by using the manufacturer’s recommended CV charge voltage. Almost all the VRLAB manufacturers recommend a starting current of 0.25 to 0.30 C amperes.
That is, for a 100 Ah battery, an initial current of 25 to 30 amperes can be selected. The higher current is used to charge deep-discharged batteries while the lower one for a normally discharged battery. The effect of a lower charging voltage is that the temperature rise will be less compared with a battery charged, with a higher current, but the time taken for a full charge will be more.
At the end of charging, the battery voltage attains parity with the impressed voltage the charging current tapers to a very low value. Universally, the current at the end may reach a value of 2 to 4 mA for each Ah of the capacity of the battery. At 2.25 to 2.3 V per cell, no gas evolution is observed in properly fabricated batteries. However, gassing will be evident at 2.4 V per cell. The volume of gas evolved at 2.4 V per cell is about 1000 ml in 40-50 minutes for 6V/1500 Ah VRLAB
According to Clause 6.1.a. of Japanese Industrial Standard, JIS 8702-1:1998, the duration of charge will be approximately 16 hours or until the current does not change by more than 10 % of 20 hour rate current (I20) amperes within two consecutive hours [JIS 8702-1:1998]. For example, if the 20-h capacity of a battery (irrespective of its voltage of the battery) is 60 Ah20, then the charge would have been completed if the current does not change by more than 300 mA (i.e., I20 = 60 Ah /20 A = 3 A. Therefore, 0.1 of I20 = 0.3A)
The details of CP charge of VR batteries are shown in the Figures
The charging efficiency is better than the constant current method. The demerit of this method is that it requires a stabilized voltage at a high current drain, which is costly. This method is used for float operation of the stationary cells for telecommunication and UPS applications.


Modified constant potential charging - Battery charger
In industrial applications, such a method is used where the charging circuit is an integral part of the system. Examples are automobiles, UPS etc. A series resistance to limit the current is included in the circuit, the value of which is maintained until a preset voltage is attained. Thereafter the voltage is maintained constant until the battery is called upon to perform its duty to supply starting current, emergency power etc.
The choice of the fixed series resistance depends upon the number of cells in the batteries and their ampere-hour capacity and on the duration available for charging. The applied voltage is kept constant at about 2.6 to 2.65 volts per cell.
As the charging progresses, the charging current starts falling from an initial value. When the voltage rises gradually up to 2.35 to 2.40 volts per cell, the gassing voltage tends to rise rapidly and hence the charging current falls at a faster rate.
The modified constant-potential charge is common for deep-cycling batteries such as traction batteries. The factories normally employ a fixed discharge-charge time profile such as 6-hour operation of the fork lift truck to a depth of discharge (DOD) of 80% and a recharge of 8 hour period. The charger is set for the gassing voltage and the starting current is limited to 15 to 20 A per 100 Ah. The current begins to taper at constant voltage to the finishing rate of 4.5 to 5 A per 100 Ah, which is then maintained to the end of the charge. Total charge time is controlled by a timer.
There are battery charger which have the provisions to keep the batteries connected to it even after the completion of charge to maintain the batteries in fully charged condition. This is achieved by providing short periods of refreshing charge every 6 hours to maintain its condition
The details are given in Figure 12 [12. Special Issue on Lead-Acid Batteries, J. Power Sources 2(1) (1977/1978) 96-98]

Combination methods (CC-CV methods) - Battery charger
In this method constant-current and constant-potential charging are combined together. This method is also known as (IU) (I for current and U for voltage) charging method. In the initial period of charge, the battery is charged at constant current mode till the battery reaches gassing voltage and then is switched over to constant potential mode. This method does away with the deleterious effect of the constant current charging method at the end of charging.
The charging characteristics of this method are shown in Figure 11 on the right.

Taper charging - Battery charger
The meaning of taper is to slope down. As the term plainly indicates, the current is allowed to reduce from a higher value to a lower one, by fixing the starting charge voltage at about 2.1 V per cell and terminating at 2.6 V per cell. The ratio of the current values at these voltages is referred to as taper value.
Thus, a charger with an out-put of 50 A at 2.1 V per cell and 25 A at 2.6 V per cell, is described as having a taper characteristic of 2:l.
There are single-step taper charging and two-step taper charging methods
Single step taper charging - Battery charger
In this type of charging, the current tapers from a higher starting value to a lower one of finishing rate, which is usually about 4 to 5% of the 20-hour rate capacity of the battery. Gassing is a required phenomenon since it helps to equalize the density gradient of the electrolyte. i.e., it neutralizes the stratification phenomenon. Hence, the finishing rate is fixed at sufficiently high value to allow this process to occur and at the same time does not unduly corrode the positive grids. Here, the charger output voltage is set at about 2.7 volts per cell initially and is made to come down to about 2.1 to 2.2 volts per cell at the end of the charging period.
The charging current is made to taper down slowly till the gassing voltage (about 2.4 V per cell) is attained (SOC = 75 to 80 %) and tapers at a faster rate thereafter. Normally, the taper ratio is fixed at 2:1 or at 1.7 to 1 ratio. The time taken for the completion of the charge is around 12 hours. The charging period after the gassing voltage is reached is controlled by incorporating a timing device that starts operating when the gassing voltage is reached.
The charging period can be reduced to 8 to 10 hours, but the starting current is to be enhanced, which cannot be done without consideration of the economics involved and the affordability of the consumer.

The charging characteristics of single step taper charging are shown in Figure 12

Two step taper charging - Battery charger
This method of charging is similar to the single step taper charging except the fact that the total charging time is reduced to about 8 to 10h. As the battery is capable of accepting charge at a faster rate when it is deeply discharged, a high current is employed in the first step till the battery reaches the gassing stage. Nearly 70 to 80% of ampere hours to be returned to the battery is given to the battery in the first step at a faster rate and the remaining ampere-hours are fed in the second step.
The charging characteristics of a 12V, 500 Ah battery by single-step taper charging are shown in the Figure 13
The taper charging methods are more popular for charging the traction batteries which are normally deeply discharged. Fleet operators of electric vehicles, for example postal delivery vans, milk delivery vehicles, require sophisticated battery charger to obtain the best possible performance from the batteries and to protect the large investment of cash involved.
Initial charge
A new lead-acid battery needs activation and this process of charging for the first time is called initial filling charging. The battery is filled with required amount of electrolyte and is fully charged before being sent for shipping. Normally this initial charging is done by constant current charging method at a low current for a long period till the battery attains a voltage of 16.5 V or more for being fully charged.
Nowadays, this process has become redundant as we get factory-charged batteries ready to use or dry-charged batteries which require only the addition of electrolyte.
Equalization charge
Equalizing charge Cell to cell differences are a fact one has to accept. No two cells can be the same in all aspects. Differences in the active material weights, minor variations in the specific gravity of the electrolyte, porosity of the electrodes, etc are some of the differences. Because of these reasons, each cell in a battery has its own characteristics; each requires a slightly different amount of charge. Equalizing charge once in a while keeps the end of life of the battery away. 12V automotive batteries are floated at 14.4V. A fully charged battery requires voltage levels of 16.5 V, which is never realized in service onboard the vehicle.
Hence equalizing charge (also called bench charging) is required to prolong the life of an automotive battery. Thus, a battery which receives periodic bench charge every six months may outlive batteries not receiving the bench charge, at least by 10-12 months. Frequency and extent of equalizing charges should be discussed with the battery manufacturer. With pre-programmed chargers an ‘equalizing charge’ is sometimes available via a switch that provides a continuous low current used to stabilize the voltage and relative density of the electrolyte of the cells.
Similarly, the UPS emergency power supply batteries and forklift truck batteries also require such equalization charges. A battery used in an inverter is charged only up to 13.8 to 14.4 V. As said earlier, this is not sufficient to equalize the imbalance between cells in a battery. These batteries, if given periodical equalization charges, will survive longer.
Batteries are to be given an equalization charge every six months. But traction batteries used in forklift batteries should be given an equalization charge once every sixth or eleventh cycle, depending on whether the batteries are new or aged. Newer batteries can be given an equalization charge once every 11 cycles and older ones every 6th cycle. If the batteries receive regular full charges daily, the frequency of equalization charges can be reduced to 10th and 20th cycles. An equalization charge shall be terminated when the cells show no further increase in voltage and specific gravity readings over a period of 2 to 3 hours.
Read a detailed article on Equalization charge here.
Opportunity charging
Where an off-road or on-road electric vehicle is being intensively operated, plugging into a charger during breaks and other brief rest periods can also help to extend the vehicle’s effective working shift and thus reduce the downtime of the EVs. Opportunity charging is the term given to such partial charging during lunchtime or rest period.
Such opportunity charges tend to reduce the life of the batteries. The battery counts such a charge and subsequent discharge as one shallow cycle. As much as possible opportunity charges should be avoided. The normal charging provides 15 to 20 A per 100Ah capacity, while opportunity charges provide slightly higher currents of 25 A per 100Ah capacity. It results in higher temperature and accelerates corrosion of the positive grids. And hence the life will be reduced.
Gas-controlled charging
The thermal conductivity of the hydrogen gas evolved is used to monitor the charging current. Hydrogen gas, a very good coolant is used to cool a heated element. The change of resistance of the heating element is used to regulate the current. A thermistor may also be used to regulate the current. Sometimes, the heating effect due to the recombination of hydrogen gas and oxygen gas evolved in the cell over suitable catalyst is used to operate a heat switch to regulate the current.
Trickle charging
In a continuous charge, the charger equalizes the losses due to self-discharge and intermittent discharge. A maintenance charge compensates for self-discharge. The two operating modes are characterized by constant terminal voltages:
Maintenance charge 2.20 to 2.25 V per cell
Continuous charge 2.25 to 2.35 V per cell
Depending on the age and condition of the battery, a current density of 40 to 100 mA/100 Ah nominal capacity may be necessary during the maintenance charge (trickle charge).
The continuous charge current depends to a large extent on the load profile. Batteries on maintenance charge must be recharged after each power outage. The same is true of batteries on continuous charge after unplanned loads.
Boost charging
Boost charging is resorted to when a discharged battery is required to be used in an emergency when no other battery is available and the SOC is not enough for the emergency work. Thus, a lead-acid battery can be charged at high currents depending on the time available and the SOC of the battery. Since fast chargers are available nowadays, boost charging is familiar today. Normally such boost chargers begin charging at 100A and tapers to 80A. The most important thing is that the temperature should not be allowed to exceed 48-50oC.
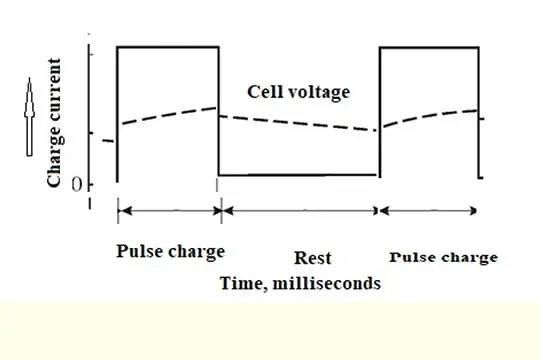
Pulse charging
What is pulsed-current charging?
Charging is done for a very short duration i.e., current on-time in milliseconds (ms), and an idle period follows (off-time in ms). Sometimes a discharge may also precede the pulse charge.
A pulsed-current technique has been applied for the rapid charging of automotive lead-acid cells. The following conclusions have been arrived at:
- The pulsed current technique can exert highly advantageous effects.
- It improves the rate of recharge.
- It has a beneficial on the cycle-life performance of lead/acid batteries, especially when an on-time of more than 100 ms is used.
- Moreover, this technique can also rejuvenate cells that have been cycled with constant current charging.
- Recharging time can be reduced by an order of magnitude, i.e., from ~10 hours to ~1 hour
- Cycle life can be increased by a factor of three to four.
- The application of pulsed-current charging to a cycled battery (capacity = 80% initial value) can evoke a recovery in battery capacity.
- Premature capacity loss occurs in both Pb-Sb and Pb-Ca-Sn cells at high rates of discharge with constant current charging.
For more details, readers can refer to the article by Lam and others given above.
Submarine cells have been the subject of pulse charging [14. Melvyn James, Jock Grummett, Martin Rowan and Jeremy Newman, Journal of Power Sources 162 (2006) 878–883 879]. The authors have concluded that
- The capacity can be improved with pulse charging. This capacity improvement
was dramatic for newer relatively new cells. But for older cells (4–5 years old) 15 or more pulse charge cycles were needed before capacity improvements were obtained.
- The older the cells had suffered severe sulphation, which takes more cycles to break down.
- Some sulphation is impossible to reverse.
- The use of pulse charging also indicated that the gassing charge could be substantially reduced.
- The gas evolution reduces with increased pulse frequency. This is more pronounced with the oxygen evolution, which is an important factor for submarine batteries that suffer from positive plate corrosion since oxygen is evolved from the positive plate during gas charging.
- After the application of pulse charging to a cell, the beneficial effects remain even though conventional charge routines are resumed.
Typical pulse charge program are shown below:
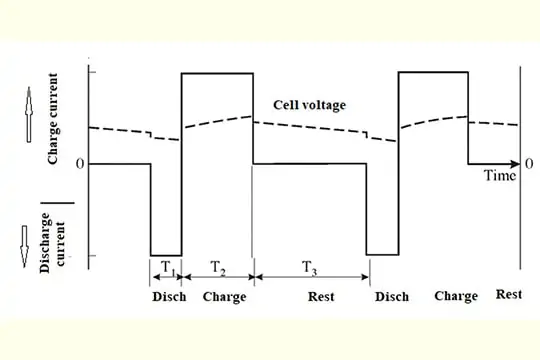
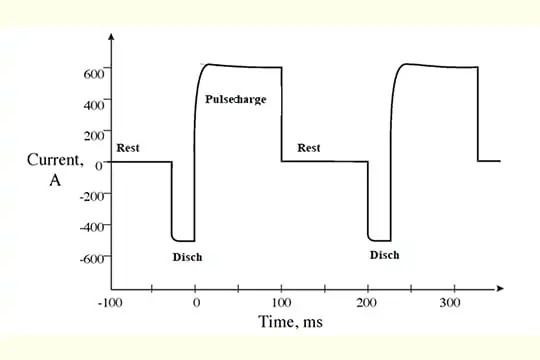
The application of pulse charging can help prevent sulfation from building up over time. It may be able to reduce sulphation buildup in cells with proper charging and maintenance if pulse charging is used from the beginning. An accumulation of sulphation that has already occurred can’t be reversed with this method. If the cells are constantly equalized or overcharged, this damages the cells, reducing their capacity and life. Microtex recommends testing the specific gravity of your batteries regularly to find out how long they will last, identify any weak or failing cells, and confirm their state of charge. The following steps can be followed in the event of sulphation buildup or an imbalance of the charge.
Fast or Rapid charging - Battery charger
Twenty-five years ago, it was believed that lead-acid batteries should not be charged at high rates because the positive active material would be irreparably damaged. It was believed that fast charging would result in excessive levels of grid corrosion and gassing, resulting in an early and rapid failure of VRLA batteries.
Rapid charge is proving to be not only time and energy saving, but it also eliminates gassing and reduces maintenance. Fast charging was first proposed by Kordesch in the year 1972 for sealed Ni-Cd cells, [17. K. Kordesch, J. Electrochem. Soc., 113 (1972) 1053] was later developed in 1993 by Norvik Technologies in Canada for VRLA batteries.
Their Minitcharger™ proved that recharge of deep-discharged Ni-Cd batteries could be achieved in 5 to 10 minutes [18. J.K. Nor, U.S. Patent 5,202,617(1993)].
In the first part of the 1990s, Valeriote, Nor, and Ettel of Cominco, Canada, advanced this technology to conventional lead-acid batteries [19. E.M. Valeriote, J. Nor, V.A. Ettel, Proc. Fifth International Lead-Acid Battery Seminar, Vienna, VA, USA, 17-19 April 1991, pp 93-122]. In the year 1994, Valeriote, Chang, and Jochim proved that the process was also suitable for thin-plate VRLA batteries [M. Valeriote, T.G. Chang, D.M. Jochim, Proc. of the 9th Annual Battery Conference on Applications and Advances, Long Beach, CA, USA, January 1994, pp. 33-38].
Since the early nineties this technique has been applied to traction batteries of all kinds [20. K. Nor and J.L. Vogt, Proc. 13th Annual Battery Conference on Application and Advances, January 13-16, 1998, Long Beach, CA, 191-197].
The effects of very fast charging on the following two types of deep-cycling hybrid lead/acid batteries were studied in 1994 using a MinitchargerÔ (Norvik Traction Inc., Canada) [21. T.G. Chang, E.M. Valeriote and D.M. Jochim, J. Power Sources 48 (1994) 163-175].
- Flooded hybrid batteries (referred to as “AP” in this work) had positive grids made from an antimonial alloy of 4.7% and negative grids of the expanded type made from high-calcium-low-tin alloy (Pb- 0.1 wt.% Ca-0.3wt.% Sn). The PAM weight was ~800 g, and the NAM ~ 540 g in each cell. It was deep-discharge type and had a capacity of 80 Ah20, 54.4 Ah5 and 50.9 Ah3)
- Valve-regulated batteries with gravity cast positive grids were made from a low antimony alloy (Pb -1.5wt. % Sb-0.3wt. % Sn (This battery referred as “ST” batteries in this work). The configuration was 5P + 6N. The negative grids were cast from Pb-O.12wt.%Ca-O.4wt.%Sn alloy. These batteries were meant for deep-cycling applications. The capacities of the batteries were 54.5 Ah5 and 52.5 Ah3
It was found that both 5-min/50%-recharge and 15-min/80%-recharge rates could be achieved, in the case of a flooded battery, with a quite acceptable temperature rise. Following an 80% depth-of-discharge, the dominant source of heat was ohmic during the first 40% of the charge returned at very high rates, 300 A (5 to 6 C3 amperes). The temperatures were distributed non-uniformly within the battery. After this, non-ohmic polarization became progressively more important. For the hybrid recombination battery, the oxygen cycle is a substantial source of heat during the later stages of the charge, particularly in comparison with previous non-antimonial batteries that have been investigated [21 T.G. Chang, E.M. Valeriote and D.M. Jochim, J. Power Sources 48 (1994) 163-175].
Fast charging of flooded and VRLA batteries
Table 6.
[21. T.G. Chang, E.M. Valeriote and D.M. Jochim, J. Power Sources 48 (1994) 163-175].]
| Flooded battery | Valve-regulated battery | |
|---|---|---|
| 5-min/50%-recharge and 15-min/80%-recharge rates | Yes | Yes |
| Temperature rise | Acceptable | Acceptable |
| Source of heat | Ohmic (up to 40 % of charge) | Oxygen cycle is a substantial source of heat during the later stages of the charge |
| Charging | charged at a constant resistance-free voltage of 2.45 V/cell (14.7 V/battery) | charged at a constant resistance-free voltage of 2.45 V/cell (14.7 V/battery) |
| Current | 250 to 300 A (5 to 6 C3 amperes) | 250 to 300 A (5 to 6 C3 amperes) |
| In the initial 3 minutes | 1 V higher than VRB | |
| Current reducing | Started to decline from the 300-A level after 3 min of charging | Started to decline from the 300-A level after 3 min of charging |
| Temperature | Higher ohmic heating and a much higher rate of temperature increase; began to decrease after 4 min | Current started to decline only after 4 minutes of charging, and it was higher than that for flooded type throughout the rest of the charging period. |
| When the current for VR battery decreased, the rate of the temperature increase became greater. After 6 minutes, although the temperature was still rising, the rate of increase began to decline. The temperature started a slow decline only after about 20 min of charging; with the same constant resistance-free voltage, VR battery accepted a higher current, which generated even more heat. The energy spent on the oxygen cycle is completely (100%) converted into heat, in comparison with about 40% for water decomposition alone. |
Figure 17. Charging: Vref=2.45 V/cell; Current, I, =3OO A max; DOD = 80%. [21. T.G. Chang, E.M. Valeriote and D.M. Jochim, J. Power Sources 48 (1994) 163-175.]
Comparison of Fast charging of flooded and VRLA batteries.

Table 7. Battery Life with MinitCharger®
[22. K. Tomantschger, E.V. Valeriote, J.S. Klarchuk, T.G. Chang, M.J. Dewar, V. Ferrone, and D.M. Jochim, Proc. 13th Annual Battery Conference on Applicationand Advances, January 13-16, 1998, Long Beach, CA, 173-178.]
| Battery Type | Battery Cycle Life | ||
|---|---|---|---|
| Conventional battery charger | MinitCharger® | Source | |
| Ni-Cd cells, type A | 500 | 1400 | INCO(1989) |
| Ni-Cd cells, type B | 450 | 1900 | INCO(1996) |
| Ni-MH cells, type A | 400 | 1600 | INCO (1996) |
| Ni-MH cells, type B | 1500 | Over 4000 | INCO (1996) |
| Lead acid Traction battery, VRLA type | 250 | 1500 | COMINCO (1997) |
Chang and Jochim have also obtained similar results. They subjected 12V VRLA batteries (spirally wound type) to conventional-charge and rapid-charge cycling tests [21. T.G. Chang, E.M. Valeriote and D.M. Jochim, J. Power Sources 48 (1994) 163-175. 23. Chang, T.G., Jochim, D.M., J. Power Sources, 91 (2000) 177-192]. The cycle life was 250 cycles for the conventional charge regime and 1000 cycles for the rapid-charge regime.
Very fast charge has been met with great success and resulted in higher life. A survey has shown that Cominco Research Team [ 22. K. Tomantschger, E.V. Valeriote, J.S. Klarchuk, T.G. Chang, M.J. Dewar, V. Ferrone, and D.M. Jochim, Proc. 13th Annual Battery Conference on Application and Advances, January 13-16, 1998, Long Beach, CA, 173-178.] conducted a survey and the team found that thirty commercially available varieties of lead acid batteries are capable of being recharged to 50% in 5 minutes, 80% in 15 minutes, and 100% in 30 minutes. In this respect, the performance of VRLAB is better than flooded SLI batteries.
Conventionally charged positive active materials are characterized by larger particles and numerous big pores. In the fast-charged plates no large particles, pores or voids were observed. The conventionally charged plates exhibited 2 m2/g surface area of PAM and those charged at high current exhibited surface area value of 3 m2/g even after 900 cycles [22. K. Tomantschger, E.V. Valeriote, J.S. Klarchuk, T.G. Chang, M.J. Dewar, V. Ferrone, and D.M. Jochim, Proc. 13th Annual Battery Conference on Application and Advances, January 13-16, 1998, Long Beach, CA, 173-178].
The PAM in the latter case expanded only slowly and consequently less pressure was exerted on the separator and the negative plate, thus reducing the risk of “soak through” shorts in the separators and NAM densification. The dramatic effect of fast charge is that on subjecting to life cycle test 12V/50Ah spirally wound VR LAB (when tested under 10h and 15 minutes charge regimes) the conventionally charged batteries could give only 250 cycles (to 80% of initial capacity) whereas those under fast charge regime could give about four times more cycles.
SEM pictures of PAM & NAM of conventional and fast charged plates

A similar result has been obtained in the works of P. T. Moseley [Journal of Power Sources 73 _1998. 122–126] ALABC-CSIRO Project No. AMC-009). High-rate battery charging of VRLA batteries restores the positive active material in a high surface area form characterized by a needle-like habit and when the battery is recharged at lower rates the positive active material forms larger particles.

Battery charger diagram




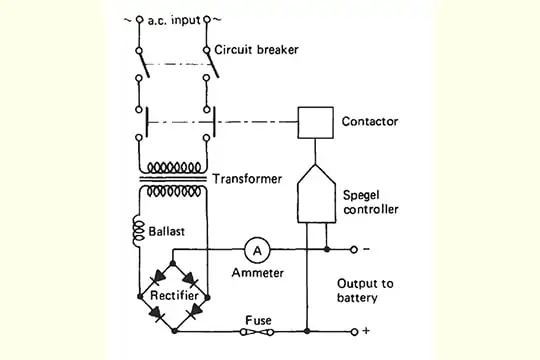

How long can you leave a battery charger on a battery?
This depends on two factors:
- Whether the charger is live or not?
- Whether there is a provision to give intermittent refreshing charge in the charger?
If the charger is switched off, there probably is no harm in leaving the battery connected to the charger, provided there is no malfunctioning of any part of the charge, like a wrong connection of AC wires leading to the chargers.
However, if the charger is switched on, the battery should preferably be disconnected so that the deleterious effects of overcharging do not reduce the lifetime of the battery.
In case there is a provision to give intermittent refreshing charge in the charger, one can leave the battery connected to the charger. This will help to maintain the battery in a fully charged condition and can be used any time the battery is required.
How does a car battery charger work?
The automotive electric system consists of the following components:
The starting, lighting and ignition system (SLI system) has both mechanical and electrical components/equipment that work in unison to crank the engine and keep the vehicle in good operation.
The major components are:
- Ignition switch
- The battery of 12V or 24 V.
- High torque DC Starter motor (or Cranking motor) with associated components
- Alternator-rectifier arrangement
- Voltage Controllers or regulators (Cut-out and cut-in relays)
When the driver turns on the ignition switch, heavy current flows from the battery to the starter motor via a control circuit and the starter motor can turn the wheels and so the vehicle begins to move.
The purpose of a starter motor is to help the engine attain some speed so that it can work. The starter therefore helps the engine to attain the intended speed to run the car. After it is done, the starter is no longer useful and thus turns off.
In automatic battery charger, a voltage sensor circuit is incorporated to sense the voltage of the battery under charge. The charger is automatically switched OFF when the battery voltage reaches the required optimum level.
Current flows along a single cable from the battery positive terminal to the component being powered, and back to the battery through the car’s metal body (which is made the earth, the negative terminal of the battery is connected to the body of the car). The body is connected to the earth terminal (negative terminal) of the battery by a thick cable.
The current supplied by the battery to the starter motor is 3 to 4 times the capacity of the battery, 150 to 400 amperes). That is, the battery supplies a current of 3C to 4C amperes to the starter motor. Therefore, the cable carrying this current should be adequately designed for the least voltage drop. The two main functions of automobile ignition system are to produce enough voltage so that it can easily create a spark for burning air/fuel mixture and secondly it exercises control over the timing of spark and transmit it to the apt cylinder. A typical automobile ignition system produces voltage somewhere between 20000 volts and 50000 volts from a 12-volt source.
Size of the battery varies with the capacity of the car. Thus, for a small car like Maruti 800 or Alto, a 12V/33 Ah battery is used, while for a Tata or Benz truck 12V or 24 V/180 Ah battery is used.
An automobile charging system generally generates a voltage between 13.5 and 14.4 volts when the engine is working. It produces electrical current for operating automobile lights, music systems, heater, engine electrical system. Long back, DC generators were used in automobiles. In the early 60s, the alternator-rectifier system replaced the DC generator because of its advantages over the other. But with advancements in electrical and electronics, all cars use an alternator-rectifier arrangement (AC is generated and converted to DC.)
In spark ignition engines, a device is required to ignite the compressed air-fuel mixture at the end of compression stroke. Ignition system fulfills this requirement. It is a part of electrical system which carries the electric current at required voltage to the spark plug which generates spark at correct time. It consists of a battery, switch, distributor ignition coil, spark plugs and necessary wiring.
A compression ignition engine, i.e., a diesel engine does not require any ignition system, because, self-ignition of the fuel-air mixture takes place when diesel is injected in the compressed air at high temperature at the end of the compression stroke.
To prevent the battery from draining off, manufacturers employ a voltage regulator / Cut-Out. It connects/disconnects the generator from the battery.
When the generator output is lower than the battery voltage, then it disconnects the generator from the battery. In contrast, when the output is higher, it connects the generator back to the battery. Thus, it prevents the battery from discharging at slow engine speeds. When the battery terminal voltage reaches about 14.0 to 14.4 V. the cut-out relay disconnects the battery from the charging circuit.
Can I start car with battery charger attached?
If one cannot start the vehicle with the existing battery, appropriate DC voltage can be supplied from a charger by attaching the charger leads as if they were the terminals of another similar battery. This is just like starting a vehicle by jump-start. Proper precautions should be taken before doing this work. Help from a professional must be sought.
What are the best chargers based on application?
Inverter Battery Charger
Inverters are electrical/electronic devices that convert the DC from batteries to AC to supply the needs of homes or small establishments. The rectifier performs the reverse function. That is the rectifier converts AC into DC. DC is the type of electric current needed to charge the battery and to operate some devices.
Home inverters normally have one or two 12 V batteries depending on the power requirements of the individual homes.
Uninterrupted Power Supply (UPS) is a similar device, but the time lag between the mains power failure and the resumption by the UPS is immediate (zero-time delay), whereas in an inverter the time delay is 10-20 milliseconds. In some production units and banks, this delay will result in huge loss and embarrassment to the customers and bankers. For example, in a home desktop computer, the screen will blackout when connected to an inverter, while you do not feel the power outing in the case of a UPS.
As we know well if the batteries are charged at voltages higher than 14.4 V per 12 V battery, obnoxious foul-smelling fumes and unwanted rotten egg smell will emanate from the batteries, apart from the formation of corrosion product around the terminals and connectors., which can be uncomfortable to the users, Hence, these batteries are not allowed to attain on-charge voltages of more than approximately 14.0 V and the preferred setting value is 13.8 V. Because of the reduced charge voltage, water loss due to electrolysis is also reduced, resulting in long intervals between two top-ups with approved water. And a full-wave rectification with filters is a good addition.
Battery charger for cars
The automobile electrical system takes care of the charging of the onboard SLI battery. As discussed under modified charging constant potential charging, the system has a resistance included in series to keep the initial surge current within the allowable limit. The maximum charging voltage is 14.0 to 14.4 V for a 12 V battery. The SLI battery being a shallow-cycled battery receives charge whenever the voltage comes down to a predetermined level.
For charging, the battery is connected to the stator of the alternator through an electronic device called a diode, which allows a flow of only in one direction, that is, current from stator to the battery and not in the reverse direction when the alternator idle.
Therefore, it prevents the unwanted discharge of the battery pack.
Cutout relay acts as a circuit breaker between the charging system and the battery when the alternator is not generating any current. It prevents the discharging of the battery in case the generator is not working or running at very low speeds.
Periodical water addition is a maintenance requirement in earlier versions of batteries. But, advanced batteries have low gassing levels and the addition of water is almost eliminated, or once in 12 to 18 months.
Battery charger for stationary applications
A stationary battery is the source of emergency power supply in several installations, where break in power supply even for a fraction of second is not tolerable. Large battery installations which are called upon only for a very short time to supply power are called stationary or standby or emergency power supplies. They are used in utility, switchgear, and other industrial environments. Such batteries are used to provide power for the initial period until they could start a generator so that it can take over the function.
Although there are several types of lead-acid batteries (Flat plate batteries, Planté batteries, conical plate batteries, etc.) and nickel-cadmium (Ni-Cd) batteries available for this application, most of the users prefer flooded type tubular stationary batteries, particularly, the OPzS type, for this purpose.
The most important feature of a stationary battery bank is the immediate supply of battery power in the case of normal mains failure. Because of this the battery must be always in a ready-to-act fully charged condition. Therefore, the charging system acquires importance. Its dependability is very crucial.
These batteries are float-charged by constant potential mode. They come in voltage groups of 24, 48, 72, 120 and 130 V. The capacity may range from 40 Ah to few thousand-ampere hours.
6 to 50 amps DC. Built-in alarms are included for high DC voltage, low DC voltage, positive and negative ground fault, and end of discharge. The industrial battery charger has digital controls and an LCD display. A number of safety features are included such as wire protection on all field terminals and full AC input and DC output protection
Simple guidelines for buying a battery charger
The following are the guidelines for selecting a battery charger:
- Know the voltage of the battery to be charged. For a lead-acid cell, for each cell, 3 volts are required for satisfactory and normal charging. Thus, for a 12 V battery, buy a charger with 20 V DC output at the terminals.
- Coming to the ampere details (i.e., current): from the battery label, find out the capacity of the battery. If the capacity is 100 Ah at a 10-hour rate, then a 10 % current output is sufficient. So, 10 A charger is suggested. But you can also go for a 15 A charger; then the cost will be higher. The advantage is that the battery can be charged within a lower time. The batteries can absorb higher currents in the initial periods. So, you can charge it at 15 A for the first 50 % input and then reduce the current to normal 10 %.
- The charger may be equipped with a digital or analogue voltmeter and ammeter. An additional facility would be a digital Ah meter. Also, reverse polarity protection may be added. This will protect both the battery and the charger.
- A full-wave rectifier with filters is good for obtaining longer life from batteries. Such a charger will produce low AC ripples and so the corrosion of the positive grids and temperature rise of the electrolyte during charging will be lesser.
- To summarize, for a 12 V/100 Ah battery, a charger rated at 20V/10 amperes with digital meters and filters with full-wave rectification and reverse polarity protection is a good buy.
Battery charger for trains
[References: Handbook on 25 kW/4.5kW electronic rectifier cum regulator unit (ERRU) of SG TL &AC coaches,) September 2019. “General Services: Train Lighting”, by Institution of Railway Electrical Engineers (IREE), Government of India, Ministry of Railways, Sept 2010.]
Wherever you go electricity is required and railway coaches are not exemptions for operating lights and fans. For Air Conditioned (AC) coaches, quite a good amount of electricity is required for running the air conditioning units fitted inside the coach.
One of the conventional methods to generate electricity is using alternators driven by the axles of the railway coaches with a battery of sufficient ampere-hour capacity connected parallel to feed the power to the coach during low voltage conditions. These types of coaches are called “Self-Generating (SG)” coaches.
In these SG coaches, magnetic amplifier-controlled Rectifier cum Regulator Units (RRUs) are used initially to convert AC output of alternator into DC and regulate/ control the DC voltage generated through the regulation of the field current of the alternator. This also prevents the reverse flow of current from battery to alternator during periods of non-generation.
This rectified and regulated DC power is used to operate the various electrical equipment and accessories inside the coach and to charge the batteries.
Lead acid batteries of 110 V / 120 Ah10 capacity are arranged from 3 cell monobloc units in the broad gauge (B.G.) coaches in underslung boxes . Four numbers of emergency feed terminals boxes for B.G. and one number for M.G. coach is provided on each end wall for interconnecting the coach to adjacent coach to receive power, in the case generation fails.
One number emergency terminal box is provided centrally on each side of under frame to facilitate charging of battery from external source. (For example, when the train is idle on Railway junction platforms). For BG AC coaches, 18 kW / 25 kW brushless alternators are used. Two such alternators are used in AC-2 Tier /AC-3 Tier / Chair Cars and only an alternator is used in the First AC coach. Batteries of 800 / 1100 Ah capacity at 10 h rating are used in I AC / AC-2 Tier / AC-3 Tier/chair car of B.G. Coaches.
Although the first train in India travelled 34 km with 400 people on 16th April 1883 from Bori Bunder (now renamed Mumbai CST) to Thane, train lighting system (TL) through axle driven dynamo pioneered by M/s. J. Stone & Co. came to Indian Railways only in 1930. Dynamo / brushless alternators driven from the axle through flat / ‘V’ belts, supply the load when the train is in motion and charges the batteries. The batteries supply the load when the train is idle on platforms and elsewhere.
Following systems for train lighting are presently in use –
1) Axle driven system working on 110 V DC supply.
2) Mid on generation with 415 V, 3 Phase generation AC 110 V utilization.
3) End on generation with 3 Phase 415 V generation and AC 110 V utilization
4) End on generation with 3 Phase 750 V generation and AC 110 V utilization
All coaches being built are having only a 110 V system. The coaches operated in 24 V systems have already been converted to a 110 V system.
The standard rating at the DC output terminals of the ERRU for different ratings of the alternator are given below:
(i) 25 kW, 130V, 193A
(ii) 4.5 kW 128.5V 35A
ERRU is mounted in the underframe of coach and designed to work satisfactorily in temperature ranging from -5 degree to 55 degrees C and 98% relative humidity. It is also designed to work in a heavily dusty area, to withstand service vibrations and shunting shocks.
The power transmission is through V-belts. Total 12 Nos. (6 on each side) and 4 nos. (only one side) of C-122 size are provided on AC and TL alternators respectively. The speed of the alternator varies from 0 to 2500 RPM. The coach wheel diameter is 915 mm when new and 813 mm when fully worn, new wheel diameter shall be considered for making calculations of the speed of the train in km/h corresponding to cut-in speed and Minimum Speed for Full Output (MFO) speeds of the alternator.
The output characteristics of the electronic rectifier cum regulator unit (ERRU) (both 25 kW and 4.5 kW) are given below:
The no-load DC output voltage is 135 V maximum, which can be set at 128 ± 0.5 V, 97 A (for 1100 and 650 Ah batteries) and 128 ± 0.5, 19 A for 120 Ah batteries) at 1500 rpm (midway between minimum and maximum speeds), the voltage regulation being ± 2 %, efficiency 95% (minimum). Voltage ripple is kept within 2 %. The load variation is 10 A to 193 A at speeds of 400 rpm to 2500 rpm (for 1100 and 650 Ah batteries ) and 350 RPM to 2500 rpm (120 Ah batteries).
For the higher capacity batteries, the voltage at 15% overload is 120 V (minimum) at 222 A, the current being limited to 230A (maximum). For a 120 Ah battery, the voltage at the overload of 40 A is set at 115 V (minimum).
The battery charging current limit is 220 A for 1100 Ah battery, 130 A for 650 Ah battery and 24 A for 120 Ah battery (maximum). The last two parameters can be set from Universal Voltage Controller (UVC) as well as from Coach Indication Panel (CIP).
For the 4.5 kW EERU, the load variation will be 1 A to 37.5 A at 350 RPM to 2500 rpm. The voltage at the overload of 40 A is 115 V (minimum), the current being limited to 43A (maximum).
We can see that the charging current is 1100/220 = 5; 650/130= 5 and 120/24 = 5. That is charging current is limited for all these batteries is C/5 amperes, the maximum voltage being 128 V. (i.e., 16 % above the OCV of the battery bank).
For more details on block diagrams for The Overall Coach, the wiring will be like the following diagram and Block Diagram of Alternator-ERRU System, the link given below can be referred:
Traction battery charger
The performance and life of Forklift batteries are influenced by the traction battery charger and the methods of charging employed. Forklift battery charger should be selected in conformity with the voltage and Ah of the batteries.
A good Forklift battery charger
- Should limit the temperature rise while charging
- Without undue overcharging, the charger must stop supplying current to the battery at the right time
- Should have equalization charge facility (i.e., charging at higher currents).
- In case of dangerous situations, an auto-shutoff facility to be provided.
- Should be programmable via microprocessor or a PC.
- In some chargers, air agitation via thin air pipes in the cells is also provided.
The charging voltage range varies from 24 V to 96 V.
The current depends on the capacity of the battery, which ranges from 250 Ah to 4000 Ah
Traction battery charging methods
Single-step taper charging: The charger begins its work at about 16A/100Ah and the current tapers as the cell voltage rises. When the cell voltage attains 2.4V/cell, the current tapers to 8A/100 Ah and then reaches the finishing rate of 3 to 4 A/100 Ah. The charging is switched off by a timer. It may take about 11 to 13 hours (Ah input factor 1.20) for 80 % discharged batteries without air agitation.
The difference in charging time is due to the variation of the starting current, that is, if the starting current is 16 A/100 Ah, the duration is lesser and if it is 12A/100Ah, the duration is more. With the air agitation facility, the duration is reduced to 9 to 11 hours (Ah input factor 1.10).
Two-step taper charging (CC-CV-CC mode): It is an improvement over the earlier method. The charger starts with a higher current of 32 A / 100 Ah. When the cell voltage attains 2.4 V per cell the charger automatically switches to taper mode and the current continues tapering until 2.6 V per cell is reached and the current goes to a finishing rate of 3 to 4 A/100 Ah and continues for 3 to 4 hours. It may take about 8 to 9 hours (Ah input factor 1.20) for 80 % discharged batteries without air agitation. With the air agitation facility, the duration is reduced to 7 to 8 hours (Ah input factor 1.10).
Charging of Gelled VRLA batteries: (CC-CV-CC mode):
The charger starts with a current of 15 A / 100 Ah. When the cell voltage attains 2.35 V per cell the charger automatically switches to taper mode and the charger goes into CV mode at the same voltage. This takes a maximum of 12 hours. The CV step is held constant as long as the charge current drops to a limited value of 1.4 A/ 100 Ah. The second phase may last for a few hours, a maximum being 4 hours. This duration depends on the first phase duration.
High frequency forklift battery charger
Existing chargers are generally of two types: ferro-resonant and silicon controlled rectifier (SCR). They’re more affordable, but they’re less efficient as well.
Battery charger incorporating high-frequency switching power devices, e.g., The MOSFET (Metal Oxide Semiconductor Field-effect Transistor) and IGBT (Isolated Gate Bipolar Transistor) operate at frequencies much higher than line frequencies (a few kHz to a couple hundred kHz). In contrast, MOSFETs and IGBTs, with their full-on/off capability, can be controlled precisely at any moment to allow a charger to produce the desired output. SCRs are half-controlled devices with an uncontrollable turn-off.
HF chargers function as switching power supplies, which means they switch the electronic switches on and off at high frequencies (50-170 kHz).
Advantages of this HF technology include:
| High frequency battery charger | |
|---|---|
| Up to 170 kHz High Frequency | Losses from conversion are lower |
| Enhanced charging efficiency (87 to 95%) | Lower energy cost (up to 20%) due to energy saving |
| Reduced AC ripple current | Longer life due to lower temperature rise. Lower maintenance costs due to lower water loss |
| It is universally adaptable | Flooded, AGM, and gel batteries can all be charged without any overcharging or undercharging. |
| Smaller size, lighter weight, and more space saving | It has a smaller foot space and can be easily mounted on-board |
| Such chargers are available in different ranges from chargers for 24 V to 80 V batteries with a charging current of 40 to 300 A. | |
Battery charger for underground mining battery applications
Underground mining batteries are primarily deep-cycle lead-acid batteries. Typical voltage ranges between 48 and 440 V, and capacity ranges from 700 Ah to 1550 Ah.
Charging these batteries is similar to charging traction batteries. The batteries are charged at
2.6 V initially with a current of 21 A to 17 A per 100 Ah and finally 4.5 A per 100 Ah as finishing rate. Charging can be completed in 6 to 8 hours.
The batteries conform to IS 5154:2013 Part 1 (IEC 60254-2006)
Marine battery charger
Batteries for marine applications can be classified into two types. Starter batteries have thinner plates and are able to deliver large bursts of power for a short time. The other type is a deep cycle battery used for other marine applications such as electronic accessories, a trolling motor and onboard electrical and electronic appliances. Additionally, dual-function batteries perform both as SLI batteries and deep cycle batteries. Specific chargers are used for specific batteries. CC-CV mode should be used on VR lead-acid batteries.
There are also chargers that can charge up to four batteries simultaneously. All types of marine batteries, VR batteries (both AGM and gelled) as well as low-maintenance flooded batteries can be charged.
Since batteries and chargers are used in boats, they should remain dry and have ample ventilation. They should also be waterproof, shockproof and vibration-resistant and if needed completely sealed. Additionally, you should make sure the chargers have a reverse polarity protection feature and spark-proof capabilities.
Battery charger for solar applications
Due to variations in solar irradiation, the output of the SPV panels fluctuates. As a result, a digital maximum power point tracker (MPPT) is connected between the SPV panel and the battery to ensure a worry-free charging process. An MPPT is an electronic DC to DC converter designed to optimize the match between the solar array (PV panels) and the battery bank. It senses the DC output from the solar panels, changes it to high-frequency AC and steps down to a different DC voltage and current to exactly match the power requirements of the batteries. The benefit of having an MPPT is explained below.
Most PV panels are built for an output of 16 to 18 volts, even though the nominal voltage rating of the SPV panel is 12 V. But a nominal 12 V battery may have an actual voltage range of 11.5 to 12.5 V (OCV) depending on the state of charge (SOC). Under charging conditions, an extra voltage component has to be delivered to the battery. In normal charge controllers, the extra power produced by the SPV panel is dissipated as heat, while an MPPT senses battery requirements and gives a higher power if a higher power is produced by the SPV panel. Thus, wastage, undercharge and overcharge are avoided by using an MPPT.
Temperature affects the performance of the SPV panel. When the temperature rises the efficiency of the SPV panel decreases. (Note: When SPV panel are exposed to a higher temperature, the current produced by the SPV panel will increase, while the voltage will decrease. Since the decrease in voltage is faster than the increase in current, SPV panel’s efficiency is decreased.). On the contrary, at lower temperatures, the efficiency increases. At temperatures lower than 25°C (which is the temperature of standard test conditions (STC), the efficiency increases. But the efficiency will balance out over the long run.




