Are you afraid your forklift battery will fail when you need it the most?
Have you ever had a moment when you thought your forklift battery may not function through the day when you had an important shipment to be loaded? We have, too. So, we wrote this step-by-step article to give you complete control on how your forklift battery performs.
A forklift fleet in-charge, Tony, sent me an email a few weeks ago:
“I have been using forklift batteries for many years now. I keep my batteries charged regularly. I even have scheduled water top-ups every week. Yet my batteries don’t last thru the shift. What do I do?”
In this forklift battery guide, we give you a complete perspective on forklift traction batteries and how to get the best life out of your investment. Let’s read on…!
Everything you need to know about forklift batteries
- Forklift batteries are heavy and as such, they should be handled very carefully. Because it is heavy, a single person alone should never handle it. Proper training should be
imparted to the personnel concerned. - Lifting beam or overhead hoist or equivalent material handling equipment must be used when lifting the heavy battery. It is not advisable to use a chain with two hooks. This may
cause distortion and internal damage.
- It so happens in most of the industries using forklifts, that they do not worry about the forklift batteries until it begins to show the consequences of the negligence of proper maintenance. One should understand that forklift battery is more important than the forklift itself. Without a working battery, the forklift is a non-entity.
- Proper maintenance of forklift battery is a must.
- Charger and battery voltage compatibility should be ensured.
- Batteries should be charged when their DOD reaches 20 to 30 %.
- Doing away with opportunity charging helps to prolong the life of the forklift battery.
- It is best not to interrupt an on-going charge. Let it complete.
Maximize your forklift battery life
- Proper timely top-up (watering) of forklift batteries is a key to prevent sulphation and longer lives from forklift batteries.
- Timely equalization charges are instrumental in getting expected life from forklift batteries.
- While purchasing battery chargers for your electric forklifts see that they have an auto-start and auto-stop facilities. This will help in terminating the charging process when it is fully complete, saving you the trouble of stopping it at the correct moment it has finished charging.
- Follow all the precautions and safety measures as per OSHA standards.
- The proper pathway should be clearly marked for the forklifts to travel. This will avoid untoward incidents.
- The basic principles of the battery (listed below) should be known to the forklift operators so that they can maintain it in a better way.

What is the best forklift battery? Forklift Batteries Suppliers
A forklift battery supplied by a well-established manufacturer with long-standing name and reputation, and with a large network of service points and immediate availability of service personnel, is the best forklift battery.
Where is traction battery used?
The word “traction” means to pull (a load over a surface). Traction batteries or motive-power batteries are those batteries used to power heavy vehicles which move men and materials from place to place, either inside factory premises, warehouses, or outside. Such vehicles are material handling equipment like forklifts, platform trucks, stackers, pallet trucks & electrically-propelled mining locomotives. Semi-traction batteries are used in lighter application such as Electric golf carts, Boom lifts, Jacks, Automated Guided Vehicles. Floor scrubbers with the driver in the seat and electrically propelled locomotives.
Guide to forklift battery types
These vehicles may use fossil fuel or an electrochemical power source (batteries) for propelling the electric vehicle. The vehicles using batteries are powered invariably by lead-acid forklift battery packs. Lead-acid batteries are the most proven over 162 years, reliable & economical ones. Nowadays, forklift battery lithium-ion are also finding a place in this segment, yet very expensive.
Battery-operated vehicles operate silently. They are environment friendly as against the diesel-powered forklift trucks. The battery-operated trucks do not emit obnoxious gases and thus do not pollute the environment. Passenger transportation by electric vehicles, electric boats and recreational vehicles and golf carts, wheelchairs all use traction batteries.

How does a forklift battery work? How traction battery works?
The forklift battery supplies power to an electric motor in the forklift for traction purposes and also for all accessories, as in a passenger car. When the operator turns on the ignition key of the forklift, power is supplied to the electric motor and the vehicle begins to move.
As soon as the operator turns on the ignition key, electrons begin to flow from the negative terminal of the battery and reach the positive terminal. The flow of electrons is called “current”. Thus, current begins to operate the motor. This electron flow is happening in the external circuit of the battery.
Inside the battery, chemical and electrochemical transformations occur, in which ions (charged atoms or molecules) participate. The site for these reactions is called the “electrode”. In the battery parlance, the electrodes are called “plates”. The electrodes are of two types, the positive electrode and the negative electrode. There is an electrolyte to take care of the flow of ions. The electrolyte is an (electrolytic or) ionic conductor as opposed to the grids (current collectors), small parts, terminals, and the cables, which are called electronic conductors.
In the specific case of lead-acid cells, the positive plate contains lead dioxide (also called lead peroxide), PbO2, and the negative plate, metallic lead (Pb), called spongy lead due to its porous nature. Both plates are highly porous, the aggregate porosity being 50 % and 60 %, respectively, for positive and negative electrodes. The electrolyte is a dilute aqueous solution of sulphuric acid.
When the reaction occurs, the lead dioxide and lead get converted to lead sulphate (PbSO4), and in the process, the electrolyte sulfuric acid gets diluted, due to depletion of sulphate ions. The reverse reaction occurs during the charging process, when both the positive and negative active materials are converted to their original form and the sulfuric acid becomes stronger, due to the return of sulphate ions from the lead sulphate. The open-circuit voltage (OCV, no-load voltage) of the lead-acid cell is about 2.05 to 2.12 V depending on the density or the specific gravity (i.e., relative density) of the sulfuric acid solution.

When about 40 to 60 % of the active materials gets converted to lead sulphate (depending on the current drain), the voltage of the cell begins to drop faster from about 2.1 volts. Hence when the voltage of the cell nears 1.75 V per cell, the forklift has to be switched off and the battery put on charge, as soon as possible.
History of the electric forklift
| Year | Inventor | Invented |
|---|---|---|
| 1867 | Clark Company, manufacturers of axles | “Tructractor” to move materials for captive use |
| Subsequent period | Visitors saw the above vehicle and ordered them for their use | |
| 1906 | Altoona, Pennsylvania Railroad Co. | Used battery to power baggage trolleys |
| 1909 | FL truck made of steel | |
| 1917 | The Clark Company | Introduced a truck called the Tructractor |
| 1923 | Yale | Fixed forks to elevate goods from the ground and masts to take goods to heights higher than the vehicle using one-face pallets (The forerunner of forklifts) |
| 1925 | Ball-bearing included in the wheels to enhance payload more than twice | |
| 1930 | Two-face pallets introduced | |
| 1930 WW II period | The invention of two-face and stronger long-lasting pallets and standardizing them foe stacking and lifting goods. Witnessed enhanced production of such vehicles | |
| 1932 | Patent on the principle involved in hydraulic lift | |
| The 1930s | Forklifts fitted with batteries which could operate over 8 hours | |
| 1940 | Forklifts found use in every place where heavy and large goods required to be shifted, loaded, and transported | |
| The 1950s | Warehouses expanded towards the roof (up to 125 inches) to accommodate more goods in the same space, instead of expanding and building another warehouse. | |
| Higher loads created safety concerns. Driver safety cages, backrest, etc | ||
| The 1980s | Developments in operator safety and balancing techniques to prevent tipping of the load or vehicles. Several safety aspects were added | |
| 2010 | Sales of electric forklifts were almost two- thirds of the total sales of forklifts | |
| 2015 | Energy-efficient electric forklifts with regenerative braking facilities increase the time of usage. Hydraulic service brake system with replaced with ‘E-braking’, | |
| 2015 | Lithium-ion battery was introduced in forklifts in 2015 |
Although forklifts were fitted with IC engines until the beginning of the 20th century, battery-operated forklifts started their appearance thereafter. The factors favourable for the battery are:
State regulations enforcing stringer environmental laws
The increasing cost of fuels used in forklift ICEs.
Added to these are the advantages of the greener battery-powered forklifts, such as silent mode, pollution-free operation, ease of servicing due to less moving parts.
The cost of operation is also lesser.
The extensive use of forklifts was seen only from 1926, though several improvements were implemented in the design of forklifts [https://packagingrevolution.net/history-of-the-fork-truck/].
a. The centre-controlled truck
b. The counterweight of the battery was placed farther away from the fulcrum point.
c. Ways were designed to permit the whole mast to tilt forward or backwards independently of each other mechanism.
d. Welding instead of riveting made the vehicles less heavy and stronger
e. The wheelbase was undergoing continued reduction in the diameter. The designers were careful in not overlooking the safety aspects, such as stability.
In recent years, energy-efficient battery-operated forklifts with regenerative braking technology are boon to forklift users.
The introduction of standardized pallets (1930) helped increase the production of forklifts. The forklifts were designed with batteries working for a shift of 8 hours.
To start with, lead-acid batteries were used. Slowly the traction battery evolved into what it is today. Lead-acid batteries used in forklifts have different voltages such as 24V, 30V, 36V, 48V, 72V and 80V. The capacity varies from 140 to 1550 Ah.
Nowadays, lithium-ion batteries are also being fitted in forklifts. The advantages claimed by the Li-ion battery manufacturers are:
- No topping up required
- No equalization charges
- No cooling periods required
- The specific energy is threefold that of a lead-acid battery and hence, lesser weight and volume required for the battery. As a consequence, in the same space, higher capacity batteries can be placed and so downtime is less.
- The energy efficiency during charge is higher and so this results in cost savings on electricity bills.
What is meant by traction battery? What does traction battery mean?
Traction batteries are electrochemical power sources or batteries used in all kinds of electrically- propelled vehicles. Industrial material handling vehicles and passenger cars of the EV type are noted for their lower operating and maintenance costs. Moreover, they are preferred to internal combustion vehicles because of their silent and pollution-free operation for transporting people and industrial or commercial goods from place to place.
As a rule of thumb, a 2-volt battery tubular flooded forklift cell will give around 1500 at 80% depth of discharge DOD cycles at 25’C. The AGM forklift batteries VRLA design will give around 600 – 800 cycles. For this reason, Microtex recommend that the tubular flooded battery should be used for forklifts and electric MHE applications.
Basics of a forklift battery – Battery operated forklift – battery specifications
The forklift battery of the lead-acid type is similar to other lead-acid types. The design of the plates is however different & are designed to withstand the rugged forklift application.
The forklift battery uses mainly two types of plates: the more popular tubular plate & the less used, flat plate.
The forklift batteries can also be classified based on the electrolyte they use:
- Flooded electrolyte battery
- Starved electrolyte battery (AGM Valve Regulated battery) and
- Gelled electrolyte battery (Gelled VR battery)
Thus, in all the types of the lead-acid battery, the following are the same
- The positive active material is lead dioxide (PbO2)
- The negative active material is lead (Pb)
- Dilute sulfuric acid (Acid diluted with pure water)
- The energy-producing reaction is the same:
Pb + PbO2 + 2H2SO4 discharge ↔ charge 2PbSO4 + 2H2O E° = 2.04 V
The reaction voltage is also the same. The standard cell voltage is 2.04 V. What do we understand by the term “standard conditions”, When we declare the voltage of the cell kept at 25°C, at 1 bar pressure, and with the activity of the electrolyte and other materials at unit value, we call the cell voltage as “standard cell voltage.” The approximate unit activity (activity value = 1) for the sulfuric acid occurs approximately at 1.200 specific gravity.
- This value of 2.04 V is made up of two parts; (i) One from positive active material (PAM) lead dioxide (PbO2) immersed in dilute sulfuric acid solution which has a standard electrode or plate voltage of 1.69 V and (ii) the other from negative active material (NAM) lead (Pb) immersed in dilute sulfuric acid solution showing standard electrode or plate voltage of –0.35 V.
- The combination of the two plate potential values gives the cell voltage as given below
Cell voltage = Positive plate potential – (Negative plate potential)
= 1.69 – (-0.35) = 2.04
- The rule of thumb for open-circuit voltage of a lead-acid (OCV) cell is:
OCV of a lead-acid cell = specific gravity value + 0.84 Volts.
- As the above rule of thumb indicates, the lead-acid cell voltage is dependent on the specific gravity used in the cell. The higher the specific gravity, the greater will be the voltage of the cell.
- Since the sulfuric acid is also an active material in the lead-acid cell, the cell with higher specific gravity will give more capacity. That is why in some heavy-duty cells, the specific gravity is raised from 1.280 to 1.300 or more.
- The voltage of the cell decreases during discharge and increases during charge.
During charging, when the cell voltage reaches 2.4 and above, the water in the electrolyte begins to dissociate into its component gases, namely, hydrogen and oxygen. Near the end of charging the proportion of the two gases will be H2: O2 = 2:1, as in water, H2O. Because of the large difference between the actual charging voltage and the voltage of water decomposition, heat generation is significant, although the current is rather small. During discharge, due to the small overvoltage, heat generation is also small, and the effect is further reduced by the reversible heat effect that now causes cooling.
Voltage variation of the lead-acid cell during charge and discharge

- The water dissociation voltage is 1.23 V. Therefore, water in the electrolyte containing sulfuric acid and water in a lead-acid cell should begin to dissociate as soon as the cell voltage reaches 1.23 V. But the OCV itself is 2.04 V and still, the water dissociation reaction does not occur. Why? The basis of the stability of the lead-acid cell system is described below: The oxygen overvoltage (approximately 0.45V) on the PbO2 electrode is much higher than the positive plate potential (1.690 V). Hence the water will dissociate only when the positive electrode potential reaches a voltage of about 2V.
All manufacturers prefer to use pressure-die casting techniques to manufacture spines. Depending on the application, the spines are cast from special alloys. For flooded type, a low-antimony alloy with a few grain refiners like selenium (Se), Sulphur (S), and copper (Cu) are added in fractional percentages. Tin is invariably included to improve the fluidity and castability of the molten alloy and reduce resistance. The negative grid alloy is usually a low antimony alloy. Such batteries are usually called the low-maintenance type (LM type).
Barak and his co-workers reported a value of about 1.95V at a current density of 1 mA/cm2 [Barak, M., Gillibrand, M.I.G., and Peters, K., Proc. Second International Symposium on Batteries, October 1960, p.9, Ministry of Defense Interdepartmental Committee on Batteries, UK.] and Ruetschi and Cahan have given a value of 2.0 V at 3 mA/cm2 for the oxygen evolution potential on lead. [Ruetschi, P., and Cahan, B.D., J. Electrochem. Soc. 104 (1957) 406-412]. The high oxygen overvoltage of lead dioxide in the sulfuric acid solution inhibits the oxygen evolution reaction.
- Similarly, the hydrogen overvoltage on lead in the sulphuric acid electrode is also higher and has a value of -0.95V. Thus, this value is about 600 mV higher (more negative) than the OCV of the negative electrode and so hydrogen is not evolved until the negative electrode potential reaches this value of -0.95V.
Kabanov and his co-workers [Kabanov, V., Fullippov, S., Vanyukova, L., Iofa, Z., and Prokof’Eva, A. Zhurnal Fiz. Khim., 3, (1938), XIII, p.11] have reported a value of about – 0.95 V at a current density of 0.1 mA/cm2 in 2N H2SO4 solution for the hydrogen evolution potential on lead, which is slightly higher than similar values found by Gillibrand and Lomax. [Gillibrand, M.I.G., and Lomax, G.R., Electrochem. Acta, 11 (1966) 281-287].
Fortunately for the lead-acid system, the solubility of lead sulphate in dilute sulfuric acid solution is very negligible (only a few mg per litre) and so no shape change, and migration occur during discharge, thus ensuring the stability of the system during cycling.
- The reaction mechanism of the lead-acid system is explained below; During a discharge, both the PbO2 and Pb (both of which are held firmly by lead-alloy grids and are highly porous) are dissolving as Pb2+ ions (bivalent lead ions) in the electrolyte and reappear as lead sulphate and deposit very close to the respective plates. Actually, Pb4+ in PbO2 and Pb2+ in Pb dissolve as Pb2+.
- By passing current in the opposite direction during a charge, the whole of lead sulphate is converted to the original PbO2 and Pb, on the positive plate (PP), and the negative plate (NP), respectively. Of course, a little more Ah should be put in to take care of the side reactions or secondary reactions like the dissociation of water. During charge, both starting materials are lead sulphate and dissolve as Pb2+ ions in the electrolyte and redeposit as lead dioxide and lead, on the respective plates.
- The lead ions dissolve and get converted to lead sulphate, lead and lead dioxide, and such a type of reaction in which the lead ions dissolve and re-precipitate or redeposit as some other compound of lead is called “dissolution-precipitation mechanism” or “dissolution-deposition mechanism”
- The lead sulphate formed during discharge does not deposit in one place. It deposits uniformly over the entire plate surface area, in the pores, cracks, and crevices.
- The capacity obtainable from a forklift battery depends on the current drain.

What is a traction battery pack?
A traction battery pack is a complete set of the following:
- Cells with vent caps and electrolyte level indicators or sensors
- Battery Steel Tray with cell connectors
- Electrolyte level indicators
- Optional Automatic water filling system if fitted for single-point watering
with ease - Maintenance tools (good digital multimeter or voltmeter, good clamp meter for measuring the current, syringe hydrometer, thermometer, 2-litre plastic jar, funnel, filling syringes,
etc.)
What kind of batteries do forklifts use? What type of battery is a traction battery?
Forklift batteries are rechargeable secondary batteries and are specially designed for deep cycle operation under strenuous operating conditions.
- They are manufactured in high ampere-hour capacities with several single cells connected in series to obtain the desired voltage, usually 48V and higher.
- The whole pack is housed in a corrosion-resistant steel box with special coatings.
- The cell jars and lids are made from polypropylene co polymer (PPCP) & also optionally in flame-retardant PPCP grades.
- There are provisions to prevent any shorting of the cell/battery terminals.
- For the sake of convenience, automatic water top-up facilities are also available, if requested.
- Traction batteries arrive with pre-assembled charging plugs.
- The lifting eyes provided in the outer steel box are carefully balanced. This is for avoiding untoward tipping of the battery pack while loading or unloading the battery pack into the vehicle battery compartment.
Flooded Forklift battery
Forklift battery market size
Different types of lead-acid traction batteries. They can be made in different types as given below:

VR = Valve-regulated
LM = Low maintenance
LM =Lead Acid
HD = Heavy duty
There are mainly two types of plates used for manufacturing traction lead-acid batteries: the flat plate type and the tubular plate type.
Flat positive plate flooded forklift battery
The flat plate flooded type battery uses comparatively thicker plates (far thicker than the automotive battery plates, but thinner than the tubular plates) and is the least costly type, with lesser lifetimes compared with flooded type tubular plate batteries. This type of battery uses higher wet paste densities and an additional glass mat separator to improve life. These batteries require maintenance such as regular topping up of the electrolyte level with approved water and cleaning the top of the pack and terminal connections regularly to avoid the accumulation of dust and acid pools. Some manufacturers would like to call it flat plate “semi-traction” batteries. Microtex manufactures only tubular plate semi-traction batteries.
So far, we have looked at traction battery flooded, 2v battery cells. Due to the nature of their charging and operation, this design invariably requires regular topping up with water.
Tubular positive plate flooded forklift battery
The tubular flooded type battery is the best-suited one for the traction of forklift trucks. This type uses special positive plates with polyester oxide holders called tubular bags or PT Bags. These PT bags are manufactured from acid-resistant plastic materials such as polyester, polypropylene, etc. In the centre of the PT bag, there is a special lead-alloy rod (called “spine”) serving as a current collector.
The active material is held in the annular space between the bag and the spine. There are several individual bags in one pluri-tubular bag (PT bag). The number of individual bags depends on the design of the battery. It varies from 15 to 25. All the spines are connected to a common top bar of the tubular plate grid. The diameter of the spines depends on the diameter of the bag and is a design aspect to control the life of the tubular batteries. The thicker the spine, the higher is the life of the battery.

The tubular bags are tested for their acid-resistant properties at higher temperatures. The tubular structure helps in retaining the active material in place and therefore shedding of active material is very much reduced.
All manufacturers prefer to use pressure-die casting techniques to manufacture spines. Depending on the application, the spines are cast from special alloys. For flooded type, a low-antimony alloy with a few grain refiners like selenium (Se), Sulphur (S), and copper (Cu) are added in fractional percentages. Tin is invariably included to improve the fluidity and castability of the molten alloy and reduce resistance. The negative grid alloy is usually a low antimony alloy. Such batteries are usually called the low-maintenance type (LM type).
An improved Low Maintenance battery uses higher specific energy and is constructed from similar plates, but with the following modifications:
- The cell accommodates larger area plates. This is achieved by reducing the mud-space
- Has a lower volume of electrolyte, because of a reduced level of the electrolyte above the plates.
- To make up for the reduced volume of the electrolyte, the cell has a higher relative density electrolyte, up to or a little more than 1.280 specific gravity.
- Some highly improved cells use negative grids made from copper metal stretched designs with lead-plating to protect it from corrosion.
Naturally, because of the higher specific energy and higher density electrolyte, the cells have a lower life expectancy.
Some manufacturers use a specially designed plastic bottom bar with cavities which allows for positive plate growth during continuous use.
AGM VRLA Forklift battery (Absorbent Glass Mat)
Sealed Maintenance Free or SMF forklift batteries designs, either VRLA AGM or VRLA Gel types avoid the maintenance required for topping up. This becomes important if maintenance standards are poor or expensive due to the high costs of labour required to add distilled water. However, there is a shorter cycle life associated with maintenance-free designs. The lowest cycle life being the VRLA AGM flat plate design followed by Gel battery. Both are not ideal due to lower life when used in traction applications, while they do offer the maintenance-free benefit.
The AGM VRLA forklift battery is a valve-regulated lead-acid battery and requires no water top-up. These batteries employ flat plates instead of tubular plates. Here are a few differences in the construction of AGM batteries:
- The composition of the positive and negative grid alloys is different, particularly, the negative alloy, which requires an alloy with high hydrogen overvoltage to avoid hydrogen evolution.
- These batteries use a unique separator material called absorbent glass mat (AGM) which looks like thick cardboard.
- The volume of electrolyte is limited and is fully retained by the plates and AGM separator and so it is a non-spillable type. The AGM is highly porous with high absorption properties. The electrolyte is thus immobilized, and a flooded condition of the electrolyte is avoided by using a starved electrolyte design. Because of the reduced volume of the electrolyte, the density of the same is increased to make room for higher ampere-hour capacity.
- Such batteries are assembled in a semi-sealed condition with a valve that controls the internal pressure, which in turn, assists in the – “internal oxygen cycle”. The oxygen cycle referred to here, helps in the restoration of the water electrolyzed during charge and overcharge reactions.
- The oxygen gas resulting from the dissociation of water on the positive plate during charge goes to the negative plate via voids and gas paths available in the AGM and overhead space to the negative plate and gets reduced to hydroxyl ions (OH–). These hydroxyl ions react with the hydrogen ions (H+) to reproduce the dissociated water, thus eliminating the necessity of water addition which otherwise results in flooded lead-acid systems. The water returns to the positive plate.
Such batteries are particularly helpful where the maintenance procedure is slack, and the workers are not properly trained. Additionally, the topping up cost is avoided, which includes the cost of the labour and time, and materials. The temperature rise is also higher because of the inherent nature of the internal oxygen cycle, due to which the water top-up job is eliminated.
Special heavy-duty (HD) cells with air circulation:
(and also with water cooling) facilities for higher discharge currents:
As in submarine cells, the design uses air is pumped inside the cells to nullify the effects of acid stratification and sulphation. In some cells, as soon as the charging is started, the charger pumps small volumes of air into the thin tubes fitted in each cell via special plugs.
In this case, the vent plug is specially provided with an integrated air supply system. The air supply system supplies air to the pipes as soon as the charger is connected to the battery terminals, which creates a circulating air stream for agitation of the electrolyte. Before beginning the air supply, the system inspects the electrolyte surfaces for gassing. The filter in the system should be regularly inspected for the accumulation of dust and, if necessary, replaced with a new one.
(References
http ://baterbattery.com/product/ess-electrolyte-stirring-system/
Armada traction battery bolt-on -technology literature-specifications
– in regex (TAB traction cells, Slovenia)
https://www.gs-yuasa.com/en/products/pdf/TRACTION_BATTERY_2017_FINAL.pdf
https://www.gs-yuasa.com/en/products/pdf/Traction_Battery.pdf)
The benefits are:
- Due to the uniform electrolyte density throughout the height of the cell, uniform charging reactions occur over the entire area of the plates.
- Therefore, lower charging duration and lower ampere-hour input are sufficient.
- The overcharge is reduced by about 15% compared with the normal cells without such facilities.
- As a consequence, life is also improved.
- The topping up frequency is also reduced due to lower water electrolysis.
- About 25 per cent volume is needed for topping up water.
- Temperature is also kept lower and uniform.
Cooling the cells by circulating fluid around the cells is a further improvement, which will bring down the temperature rise due to higher discharge currents and higher atmospheric temperature.
Some traction battery manufacturers also supply automatic water topping-up systems to save time and labour. Connecting a tube from a small water tank kept at a higher level compared to the battery tray height allows water to flow into cells until the electrolyte level indicators/sensors reach the correct levels.
Gel Forklift Battery
The gelled VR type differs from the flooded tubular type in using all the aspects discussed in the topic on AGM battery, except that:
The plates are of the tubular type
The separator is not AGM, but a conventional type
The immobilization of the electrolyte is achieved by the use of a gelled electrolyte, prepared by the addition of fumed silica to the sulfuric acid electrolyte. The gelled electrolyte provides gas paths for oxygen transport through cracks being developed during the initial cycles.
Microtex however, does not recommend Gel batteries for forklift applications.
Characteristics of different types of lead-acid traction batteries
| Semi-traction | AGM VR | Flooded tubular | Gelled tubular | Li-iron phosphate | |
|---|---|---|---|---|---|
| Life | Low | Medium | High | High | Long |
| Cycle life (cycles) at actual operating conditions (45 to 55ºC) | ~ 300 | 500-800 | 600-800 | 700 | 2000+ |
| Cycle life to 80% DOD (cycles) at Laboratory test conditions (20 to 25°C) | 500 | 800 | 1200 to 1500 | 1400 | 5000 |
| Can be used in any position | No | Only horizontal for tall cells | No | Yes | No |
| Type of use | Lighter | Moderate cycling | Deep cycle | Deep cycle | Deep cycle |
| Topping up | Needed regularly | Not needed | Needed regularly | Not needed | Not needed |
| Cost | Least | Medium | Low | Most | More than a lead acid battery |
How does a forklift battery work? Electric forklift battery
The life of the forklift battery is defined by the number of standard deep charge-discharge cycles which it can perform until it drops to 80% of the rated or nominal capacity.
The design to the specification of traction batteries is critical in providing a long and trouble-free operation in service. In order to achieve this, there are several key aspects of the traction cell construction which ensure that they are able to stand up to the demands of the power battery cycle duty. The key components of the battery are the positive grid alloy, the active material chemistry and the method of separation and plate support.
The forklift battery is a deep discharge battery & recharging is required with high voltage over a long period. During this process, there is grid growth in the spine grid of the positive electrode. This eventually fails over a long time as the positive conductor grid gets completely converted to PbO2. Forklift batteries must use lead alloys with high-corrosion-resistant properties to resist the growth of the grid, usually called creep.
The capacity & cycle life in a forklift battery depends on very important factors like active material density & structure to ensure a stable capacity & deliver the required lifecycle.
Alongside this, the physical construction of the multitube and the internal support provide a space which collects material shed from the plates during battery cycling. This is important as capacity reduction and failure can occur from short circuit damage due to the shed active material creating a conducting bridge between the plates as the battery ages.
Are flat plate forklift batteries better than tubular plate forklift batteries?
No, Tubular plate batteries are better.
The flat plate forklift battery (or semi-traction) battery is made from thinner plates and so the life is definitely poorer. A maximum of 300 deep cycles only can be expected from semi traction batteries, whereas the tubular battery offers more than 1500 deep cycles.
Cost-wise flat plate batteries are cheaper. Such batteries can be used only where the use of forklift is occasional.
Why are forklift batteries so heavy? (Forklift Counterbalance?) forklift battery weight
The heavy load in the rear of the forklift helps to balance and stabilize the forklift in operation with loads. Heavy loads are in the front and the heavy battery at the back, (usually below the driver’s seat) acts as a counterbalance. So the forklift will not topple over under the weight of the load in front on the fork.
Forklift accidents are happening mainly due to forklift overturns, due to instability. This endangers the operator and the workers standing nearby. Such type of accident is on the top of the forklift accidents list. This is mainly due to unstable forklift loads, improper loading and unloading methods, and operating the forklift at unduly high speeds. This shows a lack of initiative for training the forklift personnel and calls for training initiatives by the management.
Are forklift batteries expensive? forklift battery price in India
You bet they are expensive! Probably the investment cost of the battery may be almost as high as 50 to 75 % of the forklift sans the battery. During the lifetime of the forklift, it may require two or three battery packs over a period of about 8-12 years. It would be prudent to purchase a traction battery from a reputed battery manufacturer having long-standing proven products with a good traction battery manufacturing experience. Incidentally, Microtex has been manufacturing & exporting forklift batteries from the year 1977! That is almost 50 years of forklift battery manufacturing expertise! Products you can rely on.
Buying & Choosing Forklift Batteries manufacturers
Selecting a Forklift Battery –
Forklift batteries near me is not the right way to search for batteries!
The important aspect is to select only the standardized types of batteries. Standardized batteries are less costly and have shorter delivery periods.
There must be compatibility of the electric motor and the battery to be selected. We cannot use batteries with any voltage. Hence, the nameplate or the tag on the electric motor is a good guide for selecting the forklift battery.
If the previously used battery is available, the nameplate will definitely guide you to the correct battery.
How to choose the best forklift battery for your warehouse?
The best way to choose a forklift battery is to contact a well-established manufacturer with long-standing name and reputation, with a large network of service points and immediate availability of service personnel.
The following point may be considered when selecting a forklift battery:
- The average ambient temperature of the warehouse
If it is a refrigerated one, it is advisable to use a little higher capacity battery or a special heavy-duty battery
How to determine if the battery is sized correctly or rated correctly for my forklift?
The nameplate on the previously used battery will give all the details of the battery. Such as the voltage, the capacity at a definite rate (usually 5 or 6-hour rates), date of manufacture, etc.
Similarly check the tag on the machine, which may give the details of the DC motor or DC voltage input required etc. These two should tally.
How to check the required capacity of the battery in a forklift where there is no nameplate?
In the absence of a nameplate on the battery tray, identifying the battery details from the coding stamped by the manufacturer on the metallic parts of the battery, such as cell connectors.
- The best way is to contact the battery manufacturer/dealer, who is the best person to assist you in this work.
- Count and scan the inter-cell connectors for stamped coding. For example, ME36/500 may indicate that there are 36 cells, or the battery is 36 volts and ‘500’ may indicate Ah capacity at 5- or 6-hour rate.
- If you have any doubt about the voltage ratings, the number of cells can be easily counted. Multiply this number by 2 and you have the voltage of the battery.
In some coding, the number of cells or voltage of the battery, the number of Ah of one positive plate, and number of the plates used are given, for example, GT 24-100-13. The first number may indicate the cell numbers or the battery voltage. The second numeral will indicate the capacity of one positive plate. Usually, the number printed lastly will be odd. Deduct 1 from this number and divide the result by two; this will give you the number of the positive plates used in one cell. Each positive plate will be 100 Ah and so in this case, [(13-1)/2] = 6 numbers of positive plates are there. So, the capacity would be 6×100=600 Ah.
When to replace electric forklift batteries? When should you replace your forklift battery?
This is something a purchase person would like to learn about!
- The forklift operator is the best person to judge it. He will experience shorter operating times of his battery-operated forklift, even though the battery receives regular charging and also equalization charge.
- The forklift maintenance team should check for its capacity at 5-hour rate after a full charge and if the capacity is less than 80 per cent, the battery has to be replaced.
- If the forklift battery is not more than 3 years old, it is a wiser decision to replace 1 or 2 faulty cells (not more, more usually indicates a different problem) and have it repaired. Leave this task to the manufacturer.
- Do not continue using a battery with low-capacity performance in service merely because it continues to deliver power for some time. The damage will get worse.
Forklift battery specifications - forklift battery weight
The National and International standards on Motive power batteries refer only to the cell sizes and do not give any specification for trays or the type of plates to be used. The battery packs for forklifts differ in the design of internal components such as plates, separators, and terminal and pillar posts. The battery trays or battery boxes will have lifting eyelets and locking arrangements for fixing in the forklifts.
The standard cell dimensions available in Asia and North America are given in the Table below:
| Cells prevalent in Asia - Overall height | Cells prevalent in Asia - Jar Height | Cells prevalent in Asia - Width | Cells prevalent in Asia - Length | Footprints of cells prevalent in North America - Narrow Cells | Footprints of cells prevalent in North America - Wide Cells |
|---|---|---|---|---|---|
| 231 to 716 | 201 to 686 | 158 | 42 to 221 | Minimum - 50.8 x 157.2 Maximum 317 x 158.8 | Minimum - 88.9 x 219.2 Maximum 203.2 x 219.2 |
Note: Dimensions are given in mm. All dimensions refer to external dimensions.
For details of bolted terminals please refer to IS 5154 (Part 2) or IEC 60254-2, latest editions.
- The battery is rated at 5-hour rate. For example, a capacity of 500 Ah at 5 rate means that the battery can be discharged at a current equal to 500/5 = 100 amperes to an end-voltage of 1.7 V per cell at 30°C.
- But different manufacturers rate their products at 5-hour or 6-hour and also give the equivalent 20-hour rate capacity.
- The voltage of the forklift traction battery packs can be obtained at different voltage ratings such as:
- 24V, 30V, 36V, 48V, 72V, 80V
What are the key questions to ask when buying a forklift battery?
Key points to be discussed with the forklift battery manufacturer/dealer.
- What is the chemistry of the battery? That is, whether it is the standard lead-acid type or a Li-ion battery type
- If it belongs to the lead-acid battery type, what is its classification, meaning whether it is flooded type, tubular traction type or flat plate type, semi-traction type, AGM forklift batteries type, or gel
battery type. - The voltage rating
- The capacity of the battery and the rate at which it can be discharged (usually C5)
- What are the special benefits of your battery?
- What is the expected life of the battery under operating conditions in terms of years?
- What are the results of the laboratory testing as per industrial standards?
- What are the effects of temperature on the performance of the battery, particularly, life? Have you tested these parameters?
- What is the relationship of life with respect to depth of discharge (DOD)?
- What are the durations obtainable at different discharge currents?
- What is the relationship between the discharge current and the per cent capacity obtainable?
- What is the relationship between the operating temperature and capacity obtainable?
- How is the battery supplied, whether it is factory charged ready to use or we need to first charge it at our end?
- Whether the battery needs a refreshing charge, and if so, at what rate? & after how long?
- What is the type of charger to be used?
- Whether the battery needs an equalization charge, and if so, what is the frequency of the equalization charge?
- What are the ways of equalization charge?
- Whether the battery needs topping up with water? If yes, what is the frequency of topping up? If, no. why it does not need topping up?
- Does it have a special alloy with lesser frequency of water topping up?
- Whether automatic topping up option is available?
- Whether vent plug is fitted with transparent electrolyte level indicators & are supplied along with the battery?
- Or is it the standard yellow flip-top plugs without indication?
- Whether state-of-charge (SOC) sensors can be supplied along with the battery?
- Whether instructions and maintenance manual are supplied while purchasing the battery?
- Whether a list of “Dos and Do not’s” is given?
Why are some traction batteries so cheap while branded ones are so expensive?
Some manufacturers use a lesser number of plates per cell and also thinner plates. These plates will hold a lesser weight of the chemicals used to make the active materials. They also may use reclaimed materials like negative plates, cell jars, acid, separators, etc. These will help reduce the cost of manufacture and so they can offer cells or batteries at cheaper rates.
Can I buy a used forklift battery? forklift battery for sale
It is not advisable to buy used forklift batteries. The seller simply clean and repaint and give batteries with 80 to 85 % capacity. As you know, 80% is the end of life. So there is no use in getting a used forklift battery or a reconditioned battery.
No, don’t buy a used forklift battery.
How to order a forklift battery? How to choose the right forklift battery?

Forklift trucks have battery containers which are standard sizes based on multiples of the appropriate cell dimensions. These sizes are also regulated for cell and container sizes expected for BS and DIN standards. Considerations when choosing a suitable battery go beyond simply choosing the right capacity, which of course is critical. Other factors which influence battery choice include:
• The make and size of forklift
• Length of operation
• Application
• Location
• Maintenance resources
We have to understand that “forklift battery” means battery and the charger included. No meaning in getting the battery without a compatible charger.
If we are replacing the battery with a new one, we can have it three ways:
- Contact the battery manufacturer, Microtex will gladly take the details necessary to calculate the size, capacity and type of battery that will fulfil all your technical and economic requirements. Why take the risk of doing it yourself?
- Contact the dealer of the forklift or the forklift battery or
- See the nameplate giving the details of the battery or
- Identifying the battery details from the coding stamped by the manufacturer on the metallic parts of the battery, such as cell connectors.
The best way is to contact a traction battery manufacturer/dealer, who is the best person to assist you in this work.
The nameplate will help you a lot in selecting the right battery if you had seen a satisfactory service from the previous battery. Find out the voltage rating and the ampere-hour capacity and the rating of the capacity.
Count and scan the inter-cell connectors for stamped coding. For example, ME24/500 may indicate that there are 24 cells or 24 Volt and 500 may indicate Ah capacity at 5 or 6-hour rate. If you have any doubt about the voltage ratings, the number of cells can be easily counted. Multiply this number by 2 and you have the voltage of the battery.
A charger manufactured or recommended by the battery manufacturer should be purchased.
The charger should also have the facility of equalization charging settings.
Nowadays, Li-battery manufacturers enumerate the advantages of their batteries, but we have to consider the huge purchase costs.
Forklift battery FAQ's - Charging Forklift Batteries
Forklift battery charging SOP’s. Battery chargers should be selected to conform to the voltage and Ah of the batteries. The chargers and the methods of charging employed have a significant influence on the performance and life of Forklift batteries.
Forklift battery chargers:
- Should limit the temperature rise while charging
- Without undue overcharging, the charger must stop supplying current to the battery at the right time
- Should have equalization charge facility (i.e., charging at higher currents).
- In case of dangerous situations, an auto-shutoff facility is to be provided.
- The chargers should be programmable via microprocessor or a PC.
- In some chargers, air agitation via thin air pipes in the cells is also provided.
- The charging voltage range varies from 24V to 96V
- The current varies for a small battery of 250Ah to 1550Ah
Forklift battery charging procedure, hazards & safety
How to charge a forklift battery?
Forklift Battery Charging area / Forklift battery charging safety / forklift battery charging station layout / forklift battery charger power requirements:
A separate area should be set apart for charging or changing batteries with all statutory regulations. The regulations, hazards involved in handing batteries, battery acid, and chargers, and safety aspects are well covered by Occupational Safety & Health Administration web site (OSHA) (See OSHA website for details https://www.osha.gov/SLTC/etools/pit/forklift/electric.html#procedure)
Only trained personnel with adequate knowledge in emergency and first aid procedures must engage in charging or changing heavy batteries used in electric forklift trucks.
The area should have overhead hoists, conveyors, cranes, or similar equipment for handling heavy batteries safely.
The racks for keeping the chargers and spaces where batteries are kept for charging should be sufficiently insulated.
Only insulated tools should be used.
Charging procedure:
- As soon as the forklift battery is received for charging, the time of receipt and (open circuit voltage) OCV readings are recorded in the relevant log sheets.
- If there is a metal cover top for the forklift battery, it should be kept open
- The events are removed and replaced loosely over the vent holes.
- Using a multi voltage forklift battery charger, the proper charger setting is selected, and the charging clips are correctly connected to the battery terminals.
- The appropriate charging current is set, and charging commenced.
- Hourly readings of the terminal voltage, specific gravity and temperature of the electrolyte are recorded with suitable measuring means.
- Charging may take about 8 to 12 hours.
- If the battery electrolyte is warm, provide a fan for cooling purpose; exposed metallic parts like inter-cell connectors help in bringing down the temperature of the electrolyte
- The final on-charge voltage may reach about 2.6 to 2.7 V per cell.
- At this stage, copious gassing can be observed in all the cells. This is due to the high rate of water electrolysis occurring at these voltage values.
- Now, the charger can be put on finishing current mode (4 to 5 A per 100 Ah)
- The gassing should be uniform in all the cells
- After continuing the charge at the finishing rate for 3 to 4 hours, charging can be terminated.
- Before switching off the charger, all reading should be recorded.
- The top of the battery must now be cleaned well, first with a wet cloth and then with a dry cloth.
- The charging clips are disconnected.
- The battery is allowed to cool down. If the battery is required urgently, and there is no time for cooling, follow the procedure described above.
- If the temperature of the electrolyte is too warm (more than 45°C) and the area in which the forklift is operated also warm (as in foundries), it is best to have two sets of batteries for one forklift where the forklift is used in busy loading stations.
Forklift Battery Charging methods:
- Single-step taper charging: The charger begins its work at about 16 A/100 Ah and the current tapers as the cell voltage rises. When the cell voltage attains 2.4 V/cell, the current tapers to 8 A/100 Ah and then reaches the finishing rate of 3 to 4 A/100 Ah. The charging is switched off by a timer.
- It may take about 11 to 13 hours (Ah input factor 1.20) for 80 % discharged batteries without air agitation. The difference in charging time is due to the variation of the starting current, that is, if the starting current is 16 A/100 Ah, the duration is lesser and if it is 12 A/100 Ah, the duration is more. With the air agitation facility, the duration is reduced to 9 to 11 hours (Ah input factor 1.10).
- Two-step taper charging (CC-CV-CC mode): It is an improvement over the earlier method. The charger starts with a higher current of 32 A / 100 Ah. When the cell voltage attains 2.4 V per cell the charger automatically switches to taper mode and the current continues tapering until 2.6 V per cell is reached and the current goes to a finishing rate of 3 to 4 A/100 Ah and continues for 3 to 4 hours. It may take about 8 to 9 hours (Ah input factor 1.20) for 80 % discharged batteries without air agitation. With the air agitation facility, the duration is reduced to 7 to 8 hours (Ah input factor 1.10).
Charging of Gel VRLA forklift batteries: (CC-CV-CC mode)
- The charger starts with a current of 15 A / 100 Ah. When the cell voltage attains 2.35 V per cell the charger automatically switches to taper mode and the charger goes into CV mode at the same voltage. This takes a maximum of 12 hours. The CV step is held constant as long as the charge current drops to a limited value of 1.4 A/ 100 Ah. The second phase may last for a few hours, a maximum being 4 hours. This duration depends on the first phase duration.
How do I correctly charge traction batteries? Forklift battery disconnect
- The first thing to do before commencing charge is to disconnect the battery from the connected loads.
- There should be a separate charging room with good ventilation. The room should also have facilities for first aid in case any acid is spilt on the skin or in the eyes. Water washing fountains for washing the eyes should also be provided.
- The chargers should be designed for charging the particular battery. Compatibility of the traction battery voltage and the charger voltage must be ensured. It is preferable to have an equalization charge setting also in the charger. The nominal voltage of a lead-acid cell is 2V. But, for charging purposes, the charger output voltage should be at least 3 V per cell.
- This is to take care of the overvoltage of the cell during charging reaction and also voltage loss due to the current conducting cables connected between the battery and charger. Thus, for charging a 48V traction battery (which has 24 cells), the charger output voltage should be equal to 3V * 24 cells = 72 V. This will also take care of the equalization charge setting.
- Connect the charging clips only to the battery terminals.
- Before commencing charge, check the level of the electrolyte. Only if the plates are not submerged in the acid, top up with water before starting the charging. Otherwise, no need to add water before charging.
- It is advisable to add water at the end of charging. This a precautionary measure to avoid flooding the top of the cells during charge. The gassing will increase the level of the electrolyte due to its volume and if overfilled, the acid from the cells will overflow and spoil the surface of the battery. This will also create problems of short-circuiting and self-discharge.
- Use only approved water or demineralized one is recommended. Do not use tap water. Tap water contains impurities which affect the life and performance of the battery. Chloride is particularly harmful. It will corrode the lead metallic parts and convert them to lead chloride, thus corroding the current-conducting grids, usually anderson forklift battery connectors, bus bars, pillar posts, etc. Iron, if present, will accelerate self-discharge.
When the cells begin to gas uniformly and vigorously, the charging can be stopped.
Intermittent charging (opportunity charging) should be totally avoided.
- Always have log sheets for charging. Record the terminal voltage readings, specific gravity, and temperature readings at regular intervals. When the voltage readings are constant for two consecutive hours, it is an indication that the battery has received a full charge.
Normally, the batteries require about 10 to 20 per cent overcharge in comparison to the previous output. Never overcharge the battery. If overcharged, the temperature of the cells will rise to abnormal values. Try to keep the temperature below 55°C.
- Specific gravity readings are temperature dependent. The temperature correction factor is – 0.007 per ten °C, eg. an electrolyte specific gravity of 1.280 at 45°C corresponds to a specific gravity of 1.290 at 30°C.
- After the charging is completed, add water to make up for the level.
- Clean the battery with a wet cloth first and then with a dry cloth.
What happens if I undercharge my traction battery regularly?
Undercharging is lethal to the life of the battery. The cell reaction will indicate that during a discharge reaction, lead dioxide (in the positive plate) and lead (in the negative plate) react with the electrolyte dilute sulfuric acid to form lead sulphate.
The overall reaction is written as
Pb + PbO2 + 2H2SO4 Discharge ↔ Charge 2PbSO4 + 2H2O E° = 2.04 V
During the subsequent charging, the lead sulphate formed in both the positive and negative plates (Double Sulphate Theory) must be fully converted back to the respective starting active materials. This is done by giving a little more of the Ah compared with the previous Ah output (10 to 30 per cent more).
If you undercharge the batteries, this conversion is incomplete, and the amount of unconverted lead sulphate will go on accumulating cycle after cycle. If the size of the lead sulphate crystals grows beyond certain limits, it is difficult to reconvert it to the respective active materials.
Undercharging should be avoided at any cost to get a good life from the forklift batteries.
This is the reason why the forklift batteries are given an equalization charge every 6th charge. This will help to completely convert the accumulated lead sulphate.
What happens if I overcharge my forklift battery regularly?
Forklift batteries require regular charging after a day’s work. This is accomplished in the charging room. The charging expert knows how to properly charge them. He knows when the forklift batteries get fully charged and when they are fully charged, he terminates the charge.
If the forklift batteries are overcharged, the temperature of the electrolyte rises to higher values than the recommended value and so the corrosion of the positive grid (and subsequent shedding or bursting of the tubular bags) will be more at a higher temperature, resulting in lower life and more volume of water required for topping up due to excessive loss of water during the overcharge. Overcharge beyond permitted levels simply electrolysis the water in the acid and the water gets split into its component gases, namely, oxygen on the positive plate and hydrogen on the negative plate.
What happens if I charge my forklifts only when I need to use them? My business is seasonal
When the forklift is used sparingly, the batteries should not be left uncharged. So, after a few partial cycles, charge the battery properly. Otherwise, next time when you want to use the forklift, you cannot start the vehicle.
A freshening charge should be given at the finishing rate (5 amperes per 100 Ah) for 3 to 4 hours if a battery has been idle for a short period. Ideally, give a freshening charge once every 4 months.
What voltage is too low for a 48-volt battery?
Under working conditions, a voltage value of 42.0 V for a 48V battery is very low. The forklift should be immediately stopped if the voltage is equivalent to 42 for a 48V battery.
Under open-circuit conditions, a voltage value of less than 48V is very low. The battery should immediately be put on charge.
Similarly, for:
| Battery voltage | Put for charging immediately if voltage is less than: |
|---|---|
| 80V | 70V |
| 48V | 42V |
| 36V | 31.5V |
| 24V | 21V |
| 12V | 10.5V |
Forklift batteries normally take 8 to 12 hours. A cooling period of about 6 to 8 hours is also needed before putting it to use. The final cell voltage may reach 2.6 to 2.65 V.
Cells equipped with air agitation of the electrolyte take lesser charging time and lesser overcharge input. They also exhibit a lower temperature rise. Life is also more. Uniform charging reactions occur over the entire area of the plates due to the uniform electrolyte density throughout the height of the cell. The topping up frequency is also reduced due to lower water electrolysis. About 25 per cent volume is needed for the topping up water.
How long should you charge a forklift battery?
Gel tubular VR batteries should be charged in a controlled manner. The charging regime is a CC-CV-CC method. The total charging time may be about 12 to 16 hours. The initial current is about 14 A/100 Ah and finishing current 1.4 A/100 Ah. The change-over voltage for CC to CV is 2.35 V.
Is it safe to leave a forklift battery charger on overnight?
Yes. Most factories charge the flooded forklift batteries overnight.
It is advisable to reduce the charging rate to that of the finishing rate (4 to 5 A per 100 Ah of 5 or 6-hour rate) when there is no supervision during the overnight charging. This will also help in avoiding excessive temperature rise and unnecessary overcharge.
A charger with an auto-shutoff is better.
When charging forklift batteries, what steps to be followed?
When charging forklift batteries, it is very important to follow the instructions in the operating manual of the forklift & the battery user manual.
- General safety precautions require that you use personal protective equipment like full shield eye goggles, rubber gloves, & nose mask.
- Remove all loose-fitting metallic ornaments like bangles or necklaces to avoid any accidental shorting.
- First, open all the vent plugs to avoid building up of pressure from charging gases.
- Check the electrolyte level in each cell, if found less, top up with demineralized water, with care not to overfill.
- Then connect the charger plug to the battery socket.
- Take readings of the cell voltages & specific gravity of all the cells at the start of the charging.
- Record the readings in the charging record (usually supplied by the manufacturer; get in touch with Microtex if you need the charging record format).
- Charge it completely for the recommended duration of 8 to 10 hours depending on the state of charge or as recommended by the traction battery manufacturer.
- Before disconnecting the charger, take final readings of the gravity to ensure it has been fully charged.
- Record the gravity.
What is the correct voltage of a traction battery cell? how to check traction battery?
What is the correct voltage of a traction battery cell? how to check traction battery?
The voltage of a traction cell depends on the specific gravity of the sulfuric acid solution inside the cell.
The rule of thumb is:
OCV (No-load voltage) = specific gravity + 0.84 Volts (in fully charged condition)
Hence, a cell with 1.250 specific gravity will have a no-load voltage of 1.25 + 0.84 = 2.09 V. Similarly, a cell with 1.280 specific gravity will have a no-load voltage of 1.28 + 0.84 = 2.12 V.
Therefore, a traction battery pack of 48 V (24 cells) will show an OCV of 2.09 *24 = 50.16 ± 0.12 V if the specific gravity is 1.250 and one with a specific gravity of 1.280 will show 50.88 ± 0.12 V
These values hold good for cells that have taken a rest period of 48 hours after charging.
A discharged cell will show lower open-circuit voltage, depending on the state of charge (SOC) or the depth of discharge (DOD).
Dependence of closed-circuit voltage (CCV) on DOD
(For a 10-hour rate of discharge)
| State of Charge (Percent) | Approximate dependence of close d circuit voltage (CCV) on DOD, Volts - Flooded Lead Acid Battery | Approximate dependence of close d circuit voltage (CCV) on DOD, Volts - Gel Battery | Approximate dependence of close d circuit voltage (CCV) on DOD, Volts - AGM Battery |
|---|---|---|---|
| 100% | >12.70 | >12.85 | >12.80 |
| 75% | 12.40 | 12.65 | 12.60 |
| 50% | 12.20 | 12.35 | 12.30 |
| 25% | 12.00 | 12.00 | 12.00 |
| 0% | 10.80 | 10.80 | 10.80 |
Note: For higher rates of discharge, the voltage values will be lower, depending on the discharge rates. The higher the discharge current, the lower will be the CCV values
The maximum charging voltages are:
Flooded Lead Acid battery 2.60 to 2.65 V per cell
AGM battery 2.35 to 2.40 V per cell
Gel battery 2.35 to 2.40 V per cell
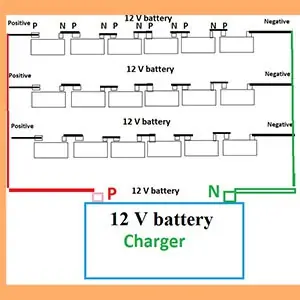
Can you charge a 36V battery with a 12V charger?
Yes, but we should not, except with the help of a trained professional.
(If possible you can convert a 36 V battery into three numbers of 12V batteries. Connect all the 12 V batteries in parallel. Be careful when connecting the cells in parallel. First, connect six cells in series (positive to negative and so on) to make a 12V battery. Similarly, make two more 12 V batteries. Now, the same polarity terminals of the three 12V batteries are connected to one current connection lead.
Now you have two leads, one positive and the other negative. You can connect the positive lead to the positive output terminal of the charger and similarly, the negative lead to the negative output terminal of the charge. Begin charging, as if it were a 12V battery. But it may take three to four times the duration of normal charging).
Arrangement of a 36 V battery into a 12V battery for charging from a 12 V charger
Equalization Charge
How to Equalize Charge a Forklift? How often should you equalize a forklift battery?
Before we discuss the equalizing charge, we have to understand the operation of forklift batteries. Most of the forklift batteries are used over an entire shift. It is very essential that the batteries must not be fully discharged or over-discharged. A maximum of 70 to 80 % discharge should only be withdrawn. The battery should not be squeeze discharged. Such over-discharging is harmful to the battery and tends to reduce the useful life.
Similarly, overcharging is also harmful. But occasional and periodical overcharging is beneficial to the battery.
Such periodical overcharging is referred to as “equalization charge”. During an equalization charge, the battery is supplied with extra energy to overcome the effects of stratification and sulphation. All the cells are brought to the same level of charge by extending the charge for a few hours more, as guided by the instructions given by the battery manufacturers. The specific gravity is also brought to the same level in all the cells.
- The batteries require an equalization charge once every sixth or eleventh cycle, depending on whether the batteries are new or aged. Newer batteries can be given an equalization charge once every 11 cycles and older ones every 6th cycle. If the batteries receive regular full charges daily, the frequency of equalization charges can be reduced to the 10th and 20th cycles.
- Log sheets for equalization charge will be helpful in knowing when the batteries attain full charge. Therefore, it is advisable to maintain regular log sheets for normal charges and equalization charges.
An equalization charge shall be stopped when the cells show no further increase in voltage and specific gravity readings for a period of 2 to 3 hours. The temperature correction for the specific gravity should also be taken into consideration. It is to be noted that the temperature correction for specific gravity is 0.007 for every 10°C change in temperature. The specific gravity readings decrease as the temperature increases and vice versa. Thus, an electrolyte with a specific gravity of 1.250 at a temperature of 20°C will measure approximately 1.235 at 40°C.
A freshening charge is used to bring a battery to a fully charged condition before it is placed in service or when it has been standing idle for a short period. It takes about three hours at the finish charge rate (3 to 6 amperes per 100 ampere hours of the battery’s 5-hour capacity rating).
The most important aspect to be noted is that the charger should have been designed for equalization charge settings. If the charger is also supplied by the battery manufacturers, it is advisable to get the same from them, for the sake of compatibility and special features.
Opportunity charging forklift batteries
Opportunity charging is the term given to the partial charging during lunchtime or rest period. Such opportunity charges tend to reduce the number of life cycles and hence the life. The battery counts it as one shallow cycle. As much as possible opportunity charges should be avoided. The normal charging provides 15 to 20 A per 100Ah capacity, while opportunity charges provide slightly higher currents of 25 A per 100Ah capacity. It results in higher temperatures and accelerated corrosion of the positive grids. And hence the life will be reduced.
Opportunity charging system
The opportunity charging system is nothing but a charger with a higher amperage capacity. This will be used whenever the forklift is not in use, for instance, during lunch recess. The charging current is a medium value between normal charging and fast charging.
Fast charging of forklift batteries: Opportunity chargers for forklifts
With a fast-charging system, forklift batteries are charged during lunch recesses, rest periods to keep the battery in ready to operate conditions. Fast-charging also requires special chargers. A fast-charged battery generally lives less than 3 years whereas a conventionally charged battery can live up to 5 years.
Fast charging is not extremely advantageous to the performance of the battery, particularly, life. Moreover, manufacturers give reduced warranty periods. Hence, the frequency of battery replacements is increased as against normal charging.
Fast charging is not suitable for all operations. But it is good for 24X7 hour operations. Fast charging does away with the need for additional batteries. Also, battery changing process between shifts is eliminated. Lesser operating space is an additional benefit due to fast charging.
With a multi-vehicle charger, multiple vehicles get charged at the same time with one AC input. The power is shared, so this is better for light-duty equipment like utility trucks, small forklifts, etc.
Are fast chargers bad for traction batteries?
Forklift batteries are charged by conventional methods for about 8 hours and should be allowed to cool down for another 8 to 12 hours. With the electrolyte agitation technique, the charging time is reduced to 8 hours with a lesser overcharge. But fast charging is accomplished in 10 to 30 minutes and charged to 80-85% SOC. The charging current is about 35 to 50 amperes per 100-ampere hours, which is more than 3 times the conventional charging current.
The following table gives the details of the three charging methods prevalent today.
Comparison of three charging methods of forklift batteries
| Conventional charging | Opportunity charging | Rapid charging | |
|---|---|---|---|
| Charging time (hours) | 8 to 12 | Depends on the available time, may be 30 minutes or more | 10 to 30 minutes |
| Is the battery to be removed from the forklift | Yes | No | No |
| Cooling after charging | Required | No | No |
| SOC when charged (%) | Almost 100 | Indeterminate | 80 to 85 |
| Special charger required | No | Yes | Yes |
| Life | Normal (Say 5 years) | Reduced | 3 years |
| Charging current | 15 to 20 A per 100 Ah | 25 A per 100 Ah | 35 to 50 A per 100 Ah |
| Exposure to heat | Normal | More | More |
| Warranty period | No change | Reduced | Reduced |
| Best Suited for | Normal operation | All types | Heavy equipment use 24X7 hours |
| Additional batteries | Required | Not required | Not required |
| Labour and maintenance cost | More | Reduced | Less |
| Charging space | Normal | Less | Less |
| Market share | 100 % | -- | Less than 10% |
Does fast charging affect the life of a traction battery?

Battery Charger Troubleshooting
Battery chargers are an integral part of the industry using forklifts. They should be inspected and maintained in working condition 24X7 hours. Only certified electrical professionals should be allowed to maintain, inspect, or repair the chargers.
If the charger is not functioning:
- Check the mains input in all the phases. It is a good practice to have indicating bulbs for the three phases. The earth wiring should also be good.
- Check the label on the nameplate and that on the charger. Both should be compatible.
- Check the output DC volts from the charger by using a good DC voltmeter.
- If not, check the miniature circuit breaker (MCB) switch, fuse, transformer, circuit board, and other components. Also, check the transformer AC voltage and rectifier output DC voltage.
- If everything is correct, begin to charge the battery and see if the voltage of the battery slowly rises. If the battery is a sulfated one, initially there will not be any rise in the voltage. Only when the high high-resistance sulphate layer has been broken, the battery voltage will rise.
- When the cell voltage reaches 2.4 V per cell, the charging current begins to taper. The charging is terminated when the cell voltage reaches 2.6 V.
- In case, the staff could not fix the trouble, call an electrical professional well experienced in battery chargers.
Forklift battery safety operation & hazards
Battery maintenance tips
Safety from hazards in charging traction batteries:
The lead-acid battery can give maximum possible life if it is properly maintained. Regular charging and periodical equalization charge help in prolonging the life of the battery.
The forklift battery should be maintained properly.
- The level of the electrolyte should be checked before putting the battery on charge.
- Water can be added before commencing a charge only if the level of the electrolyte has gone down below the top of the plates.
- Otherwise, the topping-up should be carried out only at or near the completion of charging.
- Else, it will pave way for the acid to overflow and spoil the top of the battery, reducing the performance of the battery.
Only the required volume of water should be added.
- A proper charger should be used for charging.
- The manufacturer/dealer should be consulted for this purpose.
- Good housekeeping is essential in a place where charging is carried out. The room should be properly ventilated to avoid the accumulation of hydrogen gas which will combine with oxygen with explosive violence if its volume exceeds 4%.
- The batteries should neither be overcharged nor undercharged. Both ways, life is reduced. Therefore a full charge is needed every cycle.
- Undercharging will tend to accumulate sulphate crystals leading to irreversible sulphation and thus reduce the efficiency of the forklift battery.
- Overcharging will reduce the life of the forklift battery by inducing more corrosion on the positive spines, leading to the premature end of useful performance.
- Over discharging to almost zero per cent State-of-Charge (SOC) will make the subsequent charging difficult and may require unduly longer charge times resulting in higher corrosion and reduced life.
- No metallic parts should be placed on the top of the battery. This may short circuit the cells and the danger of explosion and fire will be created.
- Lead-acid battery contains dilute sulfuric acid as the electrolyte and the terminals of a conventional battery and the external parts such as container, inter-cell connectors, covers, etc. get some sort of acid spray and also get covered with dust. Hence it is necessary to keep the external appearance neat and dry.
- The terminals should not be unduly strained by over-tightening the bolts and/or nuts.
- Tighten all bolts to specified torques as shown on the forklift battery
- The terminals should be kept clean by applying a thin layer of white petroleum jelly periodically so that no corrosion occurs between the terminals and the cable connected to it.
Smoking or use of naked flame in the battery charging room is highly dangerous and should be totally prohibited.
- Never bring the battery near a naked flame or short circuit the terminals of a battery.
- Never use more than four battery groups in parallel. If not possible to avoid such a condition, the battery manufacturers should be consulted.
Mixing used or new cells/batteries with different manufacturing dates and made by the different manufacturers should not be put in one string. Such a condition is liable to cause damage to the battery or to the associated equipment.
- Dusting by ‘cloth duster’ or cleaning by dry cloth (particularly synthetic fibre textile) must be avoided, as they will generate static electricity which may cause an explosion under certain conditions.
- The forklift battery should be charged only when it is 70 to 80 % discharged. Opportunity charging (partial charging during lunchtime or rest period) is an unwanted habit leading to reduced life of the battery. The forklift battery considers it as one cycle and hence reduces the cycle number and hence the life it can offer.
- As far as possible try to keep the operating temperature of the battery below 45°C by providing space around the battery trays. While near the end of charging, the temperature should not be allowed to exceed 55°C
Forklift battery FAQ’s – Forklift Battery Acid
Pure battery-grade sulfuric acid diluted to the required specific gravity with pure water is the electrolyte used in forklift batteries.
Normally the specific gravity value of 1.280 to 1.290 at 27°C is used in forklift traction batteries. For high-performance batteries, the specific gravity value may be higher, 1.310 specific gravity.
How much sulfuric acid in a forklift battery?
Forklift batteries are supplied factory charged with sulfuric acid usually of 1.280 specific gravity. The level of sulfuric acid inside the battery is usually 40mm above the separator guard. The sulfuric acid is the electrolyte in the cell and forms what is generally referred to as the third active material. The other two are the Positive active material & the Negative active material. Purity of the sulfuric acid plays an important role in the life & performance of the battery. Each forklift battery has a specific design volume of sulfuric acid usually forming 10 to 14 cc per ah of battery capacity.
It is very important that the end-user does not add any further acid to the battery. Only demineralised water shall be used for topping up of the cells. Care should be taken to ensure not to overfill the cells as the spill will be acidic & corrode the steel tray, causing ground shorts & damage to costly electronics in the modern forklifts.
What would happen if I touched battery acid?
The dilute acid use in traction batteries (relative density about 1.280 to 1.310)) does not do any harm, if it comes into contact with human skin. The skin should be immediately washed with plenty of water. Cotton clothes would be destroyed.
But concentrated acid is dangerous. It will create burns on the skin.
- It is dangerous if it splashes in the eyes.
- A fountain of water (available with personal safety suppliers) should be available in the factory to wash the eyes with a lot of water for a long time.
- Immediately consult an eye specialist professional.
- In case the fountain of water is not handy to use, a laboratory wash bottle for flushing the eyes with cool and pure water.
- If the acid is spilt on cotton clothes, the spot will be easily disintegrated, and a hole will appear soon. Hence, dresses made with synthetic, acid-resistant fibres must be selected.
Do forklift batteries need distilled water?
Yes. Like any other flooded type lead-acid battery, forklift battery also requires topping up with pure, approved water, if it is a conventional flooded battery. This is because of the loss of water occurs due to the water dissociation reaction taking place during charging after a certain voltage level.
To start with, there will be no gassing until the cell voltage reaches a value of 2.3V per cell (VPC). The gassing will be more at 2.4 VPC and it will be vigorous after 2.5 VPC.
The reactions occurring can be shown as:
2H2O (from the dilute electrolyte) = O2 ↑ + 2H2↑
In a conventional flooded cell, both the gases will be vented to the atmosphere (indicated by the upward arrows). This necessitates good ventilation of the charging room. Otherwise, the accumulation of hydrogen gas beyond 4% by volume will be dangerous, and explosion may also occur.
The main cause for an explosion in or near a battery is a creation of a “Spark”. A spark can cause an explosion if the hydrogen gas concentration in the battery vicinity is about 2.5 to 4.0% by volume. The lower limit for the explosive mixture of hydrogen in air is 4.1%, but for safety reasons hydrogen should not exceed 2%. The upper limit is 74%. A heavy explosion occurs with violence when the mixture contains 2 parts of hydrogen to 1 of oxygen. This condition will prevail when a battery is overcharged with vent plugs tightly screwed to the battery.
Please remember that no overfilling of the cells with water and no overcharging beyond a limit are permitted.
How do we add water to the electric forklift battery?
As in the case of other flooded lead-acid battery types,
- water can be added manually to each cell using a filling syringe or water taken in a plastic jar. Usually (like in Microtex forklift battery) each cell has an electrolyte level indicator built into the vent plug.
- While adding water utmost care should be exercised not to overfill the cells.
- Overfilling will flood the top of the battery, resulting in dilute acid seeping into the battery tray and creating corrosive atmosphere and ground shorts, if not properly insulated.
- In the absence of an electrolyte level indicator, a small glass tube (15 cm tall and 5 mm diameter) open at both ends can be used.
- Close one end with the index finger and insert the open end into the cell. Now electrolyte will fill the tube to a height of the electrolyte present in the cell. As a rule, the electrolyte level is about 30 to 40 mm above the separators. If the height in the glass tube falls short of this height, water should be filled up to the required level. Measure the volume of water added to one cell and it will be a good guide for other cells.
- Some manufacturers supply automatic water filling systems with the required one-way valves, connectors & water tubes. It is easier to use such a system. It reduces labour and also shortens the top-up time. Connecting a tube from a small water tank kept at a higher level (10 to 15 feet) to the battery tray height allows water to flow into cells until the electrolyte level indicators/sensors reach the correct levels.
- The valve in each cell permits the flow of water into the cell and the level indicator float closes the valve when the proper level of electrolyte is reached. A built-in flow indicator in the water supply pipe controls the top-up process. During filling the water flow causes the flow indicator to rotate. When all the plugs are closed the indicator shows that the filling process is complete.
In winter (when the temperature is below 0°C), batteries should only be charged or topped up in a charging room with heating arrangements.
What happens if lead-acid battery runs out of water?

Charging batteries with water below the plate can lead to shorts & fire.
The most important performance aspect of the lead-acid battery is that it operates with three active materials as against two in most other cases.
Without dilute sulfuric acid electrolyte as the ionic conduction medium, the lead-acid battery cannot function.
If the acid is totally absent in the cell, the cells cannot function. The forklift cannot be run. In cells with partially immersed plates, the output capacity will be proportionately reduced. There is also a risk of overheating & shorting of the electrodes.
Here comes the importance of the water addition, which is the most important aspect of maintenance (technically called “topping up”). This will compensate for the reduction in the level of the electrolyte caused by the charging process, particularly, near the end. When a charging cell attains a voltage above 2.4 V, gassing begins, and it will be copious when it reaches more than 2.5 V per cell.
The importance of watering a forklift battery. What happens if lead-acid battery runs out of water?
How to correctly water your forklift battery
The lead-acid battery is very well known for its property of losing water during charging, particularly, during charging beyond 2.4 V per cell. This is due to the instability of water at high voltages, its theoretical dissociation voltage being 1.23 V. However, it does not get electrolyzed at this voltage and that is why the lead-acid system is stable even beyond this voltage.
- Both the electrodes (plates) possess very high over voltages for the respective gases evolving from water, namely, oxygen from the positive plate and hydrogen from the negative plate during charging. Water splits into its component gases, namely, hydrogen and oxygen. Near the end of charging oxygen and hydrogen gases evolve on positive and the negative plates, respectively, in the ratio of 1:2.
Topping up or watering a forklift battery is of utmost importance.
- The alloys play important role in controlling the gassing voltages. High-antimony alloys promote earlier gassing, while lead-calcium alloy and low-antimony alloys delay the evolution to higher voltages. Whatever alloy is used, electrolysis of water occurs, and the volume lost has to be replaced with pure water, which, in battery parlance, is called “topping up”. If this step is not followed, the level of the electrolyte slowly goes down and in extremes cases, the plates are exposed to the atmosphere and become dry, thus disabling a part of the active materials from taking part in the energy-producing reactions, because of the non-availability of sulphuric acid electrolyte.
- In addition, the lead sulphate already present in these semi-dry parts of the plates cannot be converted to the respective active materials during charging and so sulphation occurs, as evidenced by the white streaks in these parts of the plates.
- The inability of the active materials of these sulphated portions of the plates to take part in the cell reactions shortens the operating duration of the forklift and soon the forklift would require a new battery.
What are forklift battery water filling systems?
Some manufacturers supply automatic water filling systems with the required paraphernalia. It is easier to use such a system. It reduces labour and also shortens the top-up time. Connecting a tube from a small water tank kept at a higher level (10 to 15 feet) to the battery tray height allows water to flow into cells until the electrolyte level indicators/sensors reach the correct levels.
The valve in each cell permits the flow of water into a cell and the level indicator float closes the valve when the proper level of electrolyte is reached. A built-in flow indicator in the water supply pipe controls the top-up process. During filling, the water flow causes the flow indicator to rotate. When all the plugs are closed the indicator shows that the filling process is complete.
Can I add battery acid to a traction battery if it is low?
Throughout the life of a lead-acid battery, there is no necessity for the user to add additional acid, whatever the type of the lead-acid battery may be.
However, if you know that a part of the electrolyte has been removed or spilled from the cells, we can add an equivalent amount of acid of the same specific gravity, in a fully charged condition.
This is so because the acid never goes out of the cells. Only water in the dilute acid gets split into hydrogen and oxygen during charging, for which regular topping up with water is sufficient. This is best done by the manufacturer who can ensure this operation is done environmentally safe way. The battery manufacturer is required to have the necessary infrastructure to handle battery acid & acid spill.
Can you add acid to a battery?
Acid should never be added to the battery throughout its life. The battery owner will never have to add acid in the battery. Batteries consume water during operation of the battery. The charging of a battery leads to consumption of water, present in the electrolyte, which is made up of sulfuric acid and water. The battery user should only top up this lost water which is the normal mode of operation.
When the level of electrolyte is found to be less, it will be good for the battery, to top up the level with pure DM water.
Never add acid. This will reduce the life of the battery.
- Some battery users top up the battery with acid when the battery is discharged.
- This acid addition increases the voltage & the user feels he has charged the battery.
- Sadly, this hastens the death of the battery.
- Never add acid to a battery, only water should be added.
Unless it is reliably learnt that acid has been spilt from the cells due to some reasons. If necessary, the same specific gravity acid as in a fully charged cell can be added to make up for the level.
Battery maintenance, testing & troubleshooting
Five Simple Steps to Battery Maintenance
To keep your forklift battery always ready for operation, follow these simple 5 step formula:
- Charge the forklift batteries regularly and properly
- Never miss the equalization charge (every 11th or 5th charge for new and old batteries, respectively)
- The electrolyte levels should be checked, and the specific gravity readings recorded in the log sheet, every month
- If needed, DM water should be added to the correct level as indicated by the level indicator
- The temperature of the electrolyte also should be recorded along with the specific gravity readings and the temperature must be kept lower than 45°C while the battery is providing power to the forklift. During charging, the temperature should not be allowed to exceed 55°C
Guide to Forklift battery maintenance checklist:
FOR THE FORKLIFT OPERATOR
- Check if the top of the battery is clean and dry.
- Check the terminal for any loose connections, and if not, properly tighten them
- Before switching on the forklift, check the temperature of the battery electrolyte and if it is high (more than 45ºC), do not operate the forklift. Let the battery cool down to less than 40ºC.
- While operating the forklift, see that the battery is not over-discharged.
- Stop the forklift when the State-of-charge (SoC) indicated is less than 30 %.
Do not resort to opportunity charging.
CHECKLIST FOR THE FORKLIFT SERVICE PERSON
- Carefully change/unload the battery from the forklift and follow all OSHA-mandated precautions.
- Check the level of the electrolyte and if the plates are not fully submerged in the electrolyte, add water.
- Choose the correct charger.
- Follow all the precautions while charging
- Top up if necessary, after finishing the charging.
- Never add acid for the top-up.
- Use approved water only for the top-up.
Proper Battery Care and Maintenance of Forklift Batteries
A properly maintained battery will give a trouble-free and anticipated life
- The first and foremost step is to keep the top and sides of the battery tray clean and dry. During the maintenance process, acid or water might have been spilt and it should be immediately wiped with a cloth soaked in baking soda solution and then with a wet cloth and finally with a dry cloth or cotton waste.
- Do not keep metallic tools on the top of the battery.
- Maintain log sheets for all the work done, especially, the periodical terminal voltage, specific gravity, and temperature reading. This will help a lot to trace trouble.
- Charging should be done as per the instructions provided by the manufacturers.
- While charging, vent holes should not be kept open. Vent plugs should not also be screwed down. They should be placed loosely over the vent holes so that acid spray does not spoil the top of the battery
- The electrolyte temperature must not be allowed to exceed 55°C during charging and 40°C during the operation of the forklifts.
- Equalization charge is a must for every 6th or 11th charge, depending on whether the batteries are old or new. Newer batteries, every 11th charge, and older batteries every 5th charge
- Batteries should never be overcharged

- Similarly, the batteries must not be over-discharged even though it is possible to run the forklift.
- As soon as the specified duration of the forklift operation is over, the forklift should be returned for battery changing or charging.
- Employees doing the charging operation should wear proper protective clothing, gloves and glasses.
- They should also have all the necessary tools for maintenance work. Maintenance tools are a good digital multimeter or voltmeter, a good clamp meter for measuring the current, a syringe hydrometer, a thermometer, a 2-litre plastic jar, a funnel, a filling syringe, etc.
- If there is trouble in starting the forklift, the first thing to do is to check the battery cables and connectors for their proper connections. A cable might have come loose during continuous operation or the service personnel might not have reconnected them properly after a charge, or a cable may have become worn out or become pitted due to constant use
- Check the specific gravity in each cell. The readings should be 30 points plus or minus the average specific gravity values. If abnormal variations are observed, the battery may need an extended charging.
- Similarly, check for the total voltage and individual cell voltages.
- The normal OCV 2.14 ± 0.03 V (for cells with 1.300 specific gravity).
- It is good to know the voltage readings under load, which will give a better understanding of the state of the cells.
- Cells that exhibit far lower voltage readings should be checked for the second time and if a cadmium reference electrode is available, record the cadmium voltage readings.
- Cells which show positive cadmium readings far less than 1.8 V and the negative cadmium readings far more than 0.15 V are labelled as defective.
- If the battery pack is less than three years old, repairing the cells or replacing them is advisable.
Forklift battery routine battery maintenance procedure
The currently available deep cycle forklift batteries can easily deliver 1000 to 1500 cycles at 80 % DOD. Therefore, a battery used fully on daily basis may live for 4 to 6 years. If the battery is to have a healthier life, proper maintenance is essential to get the expected life. Whether your battery is healthier or not depends on the care and maintenance you provide to the battery throughout its life.
Routine steps for battery maintenance are
- Properly charging the battery
- Proper top up with pure water whenever necessary
- Keeping the top of the battery clean and dry, without any spilt acid or accumulated dirt.
- Maintaining log sheets for all the reading of terminal voltage, specific gravity, and temperature.
Forklift Batteries Maintenance suggestions
- The battery should be kept clean and dry. While charging, the vent plugs should be loosely placed over the vent holes and should not be screwed down. This will avoid acid spray during the charging process.
- While connecting the battery terminals to the forklift or the charger, take care to see that the appropriate terminal is connected, positive to positive, and negative to negative.
- Check if all the connections are secure.
- The charging room should be well ventilated.
- Avoid sparks and flames in or near the charging room.
- Disconnect all loads while charging the battery.
- Record all the voltage, specific gravity, and temperature readings in a log sheet
- End of charge is indicated by the readings being constant for at least two consecutive readings.
- Equalization charge must be a routine affair every 11th cycle for newer batteries and every 6th cycle for batteries older than 2 years.
- An eye-wash fountain and other plumbing facilities should be easily reachable.
- Do not over-discharge the forklift battery, simply because it can run the forklift.
- Similarly, avoid overcharging.
- By avoiding overcharging, you avoid an abnormal rise in temperature of the electrolyte, which will reduce the life of the forklift battery.
- Routinely check the individual cell voltages and specific gravities of all the cells. This will give you a forewarning for equalization charge or improper charging and also adjusting the level of the electrolyte.
- Do not place any metallic tools on the battery.
- For further details see https://www.osha.gov/SLTC/etools/pit/forklift/electric.html
How to replace forklift battery?
- Any work you do on the forklift battery should be carried out with caution and with all safety measures.
- Safety and other protective equipment like acid-proof aprons, goggles, face shields should be worn by the staff
- The area is well ventilated.
- Have an acid-collection system in place for your floor area and washing soda or baking soda handy if acid spills on the floor.
- Establish an eye-wash station within a short distance of the battery changing area.
- When needed to remove the battery from the forklift, the first step is to switch off the forklift power supply from the battery.
- Trained professionals only must do the changing of the battery.
- The forklift should be firmly stopped using chocks, and the brakes applied before the battery is removed for charging or changing.
- Lifting beam or overhead hoist or equivalent material handling equipment must be used when lifting the heavy battery. It is not advisable to use a chain with two hooks. This may cause distortion and internal damage.
- Smoking is prohibited in the battery changing/charging area.
- Precautionary steps should be taken to prevent open flames, sparks, or electric arcs in battery charging areas.
- If the battery is older than 4 to 5 years, it is better to replace with a new one. The cost of repairing may not be worth the lifetime a reconditioned old battery may offer.
- However, replacing 3 or more cells is not advisable.
- Any power issues with the forklift should also be checked and corrected before deciding on the repair or replacement. A good battery may not work properly with a forklift that has power issues
- In some cases, the cost of repairs will be worth the trouble and money. Only a good battery can be repaired back to good working condition,
- An acid-resistant carboy tilter or siphon for handling acid from the old battery must be handy.
- The replaced battery is properly sitting and secure in the forklift before operating the equipment.
- Attach the positive clamp (+ usually coloured red) to the positive terminal first and then the negative clamp (- usually coloured black) to the negative terminal, checking for the proper polarity
- Tools and other metallic objects must never be left on top of forklift batteries.
How to calculate the capacity available in a traction battery?
Relationship between the current drain and the Ah obtained (Example: 500 Ah5)
(At the same temperature of 25 to 30°C)
(Ref: Indian Standard IS 1651:1991, Reaffirmed in 2002)
| Rate of discharge (hours) | Rate of discharge (amperes) | Capacity obtainable (Ah) | Percent based on 5 h capacity percent) |
|---|---|---|---|
| 5-hour rate (Rated capacity) =500 Ah | 500Ah/5 hour = 100 amperes | 500 | 100 |
| 3-hour rate (85 % of C5) = 425 Ah | 425Ah/3 hour = 142 amperes | 425 | 85 |
| 2-hour rate (75 % of C5) 375 Ah | 375 Ah/2 hour = 187 amperes | 375 | 75 |
| 1-hour rate (60 % of C5) – 300 Ah | 300 Ah/ 1 hour = 300 A | 300 | 60 |
| The same battery can deliver 600 Ah (120 % of C5) at 10 h rate and 690 Ah (138 % of C5) at 20-hour rate. |
- The capacity obtainable from a forklift battery depends on the temperature of the electrolyte. There is a decrease of approximately 5 % for every 10°C decreases in temperature. Thus the 500 Ah battery, if rated at 25°C, can deliver only 90 % capacity at a temperature of 15°
- The temperature coefficient of capacity for a flooded tubular battery is different for different temperatures (Ref: Indian Standard IS 1651:1991, Reaffirmed in 2002), but we can take the value approximately as 0.5%/°C for discharge rates from 5-hour rate to 10- hour rate.
- Similarly, there is an increase in capacity at elevated temperatures at the same temperature coefficient of capacity.
This reflects badly on a forklift battery performance operating in an air-conditioned environment of a food material storage warehouse. The lower temperature lowers the available capacity (and thus shortens the operating duration of the forklift).
How to test a load of a forklift on the battery during use?
It is also necessary to ensure your safety while performing the DC (current) measurement.
The current in amperes indicated by the clamp meter is multiplied by the voltage of the battery (on load) to get the power the electric forklift is drawing.

A clamp meter can be used to measure the DC (current) flowing in the cables carrying the current from the battery to the electrical circuit. The indicator should be kept in the DC ampere range and the clamp is held to the cable.
It can be used like a multimeter and other current measuring device; it is more convenient and even safer to use because you don’t have to break the circuit before you get a reading. To measure the current flowing through your circuit, it is more than just selecting DC Amps, opening the jaws of your clamp meter, closing it around a wire, and viewing the reading.
I am having ground leakage voltage on the body of my forklift battery; how does this happen? How to correct this?
The ground leakage is due to careless topping up, adding excess water, making it overflow along with the acid from the cells and corrode the steel tray, gradually.
- It is repeatedly stated in all the literature on forklift batteries that the top of the battery should be kept dry and clean. Overtopping up will result in dilute sulfuric acid to run into the battery tray and also between the cells. The battery tray will be corroded. Even though the steel tray is given acid-resistant coatings, a weak spot or a break in the coating will be sufficient for the acid to find a way.
- The more often the overtopping up occurs, the sooner the tray corrodes and the more severe will be the ground short. This will result in a voltage drop. Two significant ground shorts can produce an external short through the cell jar. As a result, some or all of the cells continually discharge. As the current -carrying ability of the multiple grounds increases, further complications such as jar leakage, overheating, cell failure, etc., can occur. Moreover, ground earthing can also create serious problems or failures in the electronic controls and electrical components of the vehicle.
- To prevent such problems, the top and sides of the forklift batteries should be cleaned before the accumulation of dampness or acid becomes serious. Hence it is a good practice to clean the top of the cells and battery every time the top-up is done.
- If not cleaned, although the water in the electrolyte will evaporate, the highly concentrated acid solution remains and gives the appearance of dampness.
- It will never dry up because sulfuric acid is hygroscopic in nature. When water vapour is adsorbed on a layer of sulfuric acid, the water molecules remain on the surface of the acid and are not allowed to evaporate.
- The ground short can be detected by using a good voltmeter with high input impedance, preferably a digital voltmeter.
- Connect the positive lead (red in colour) of the voltmeter on the positive terminal of the battery and touch the negative lead (black in colour) on the spot of the steel tray where bare metal is visible.
- Make sure that the negative lead is firmly in contact with the steel tray.
- Move the positive probe from one inter-cell connector to the other inter-cell connector until the lowest voltage reading is found. Now we have identified the grounded cell. Clear the short circuit path by cleaning the top of the battery with a cloth soaked in the baking soda solution, then with a wet cloth, and finally with a dry cloth. This will remove the spilt acid and corrosion product.
If the problem still persists, resealing the battery with a proper sealing compound is suggested, or replace the faulty cell.
How to establish what is a good forklift battery?
Superficially speaking, we can test the forklift battery for 5 h rate or 6 h rate capacity as per the manufacturer’s instruction. If the capacity delivers more than 120 per cent of the declared value, the battery may give comparatively higher cycles.
To know whether the battery is really good, we have to ask for a third-party certification (TPC) that too from an NABL accredited laboratory (National Accreditation Board for Testing and Calibration Laboratories).
We can also request for an in-house validation report of the particular type of battery.
If you have the time and the facilities, testing as per IS or IEC standards can be carried out in-house.
To get quicker results, an accelerated endurance test programme at an elevated temperature can be adopted. For example, instead of testing at ambient temperature, life cycling can be carried out at a temperature of 40 or 55°C to accelerate the test. Results can be extrapolated.
As per the Arrhenius equation, the life of a lead-acid battery is affected by temperature [Piyali Som and Joe Szymborski, Proc. 13th Annual Battery Conf. Applications & Advances, Jan 1998, California State Univ., Long Beach, CA pp. 285-290].
Life Acceleration Factor = 2((T−25))/10)
Life Acceleration Factor = 2((45-25)/10) = 2(20)/10) = 22 = 4
British Standard 6240-4:1997 [Obsolete] gives a table (Table A.1) for the dependence
of life of lead-acid batteries on temperature between 20 and 40°C, wherein it is given that if the life is 100 % at 20°C, then the life at 40°C will be 25 %.
The test results can clearly tell whether the forklift battery is good or not.
Preventing forklift battery sulfation
The following steps will help prevent the sulphation of the plates of forklift batteries:
- The forklift battery should never be undercharged.
- The forklift battery should never be over-discharged
- The forklift battery should not be left in a discharged condition for a long time.
- Regular topping up should be done with pure water.
- The top of the battery should be kept clean and dry
You can read a more detailed article on Sulfation here in this link
Guide to forklift battery reconditioning
Before deciding on reconditioning, you should go through the following points:
- Check all individual cell voltages, both during the rest period and also when the forklift is operating. See the spread of the voltage values and record them.
- Find out the specific gravity values of all cells and record them
- If the voltage values and specific gravity values differ by more than 0.03 points, (If the normal cell voltage under rest period is 2.12 V, the abnormal values are 2.09 and still lower voltages; if 1.280 is the normal specific gravity, then 0.03 points less means 1.250 and lower values). it is an indicator that the battery requires an extensive charge.
- The battery is to be subjected to a full discharge either through the forklift or in a laboratory. Note down the hourly voltage specific gravity and temperature readings in a log sheet.
- Again, give an extensive equalization charge and record readings as before. The differences in the readings would have narrowed down and might even have become uniform and equal. Then it is an indicator that the sulphated battery has been rejuvenated. No repair or reconditioning is needed.
- If the readings still are far away from each other, trouble in the internal parts might be there.
- Now, carefully drain the acid to an acid storage carboy
- Then drill holes to the diameter of the pillar post so that the inter-cell connector (in the case of a welded inter-cell connection) can be extracted undamaged for re-use.
- Now remove the cell elements from the cell jar for examination. It is advisable to do this under the supervision of a trained professional
- In this case, the elements in cells should undergo a thorough examination for short-circuiting either under the bottom, top or on the sides. This may happen due to the shedding of the active materials and the mud space bottom being filled up by the mud and thus short-circuiting, even though the sides are protected by plastic strips.
- If the positive and negative plates are found in good condition, wash out the mud and clean the separators and the jar and replace the element as in the original cell before repairing.
- Also, look for white streaks at the top of the plates. If white streaks are found, then it indicates improper maintenance procedures, like missing top-up with water, undercharging, etc.
- How to check if the plates are in good condition? The positive plate tubes should be intact, without any signs of bursting or damage. In the case of a flat plate, no shedding is permitted. The negative plates are always flat type in any type of lead-acid battery. The negative plate should show a shiny inner active material when scratched with a nail or knife. If the active material appears sandy, then the negative group has to be replaced.
- If whole cells are to be replaced, then it is advisable to consult the dealer/manufacturer.
- Cells older than two years should not be mixed with good cells. This will affect the performance of the good cells.
- If the battery is relatively new (less than five years old) and the problem is a minor one, repairing a forklift battery instead of buying a new one could save money.
- However, replacing 3 or more cells is not a good idea.
How to bring a dead battery back to life?
Before deciding if forklift battery cells can be revived, you have to check the year of manufacture of the battery. If the forklift battery is older than 5 years, the attempt to revive it, is a waste. If the forklift battery is comparatively new, it can be revived by proper charging after filling it with sufficient water. No acid should be added.
- The first step is to clean and dry the top of the forklift battery. If the clamps are on, they should also be removed. Use a washing soda also chemically called sodium carbonate or baking soda (sodium bicarbonate) 5 % solution in water to remove the acid from the top parts, terminals and clamps. Apply white Vaseline to the terminals and clamps.
- Check for electrolyte level and make up the level with pure water. Do not add tap water.
- Allow 2 hours to soak and check the level again. Add additional water if required.
- Measure the no-load or open-circuit voltage (OCV).
- Begin to charge the battery with an appropriate charger. For a 24 V battery, the charger output voltage should be a minimum of 36 V.
- Start with 5 to 10 amperes and record all the readings of terminal voltage, current, specific gravity and temperature in a log sheet every hour.
- See if the voltage begins to rise. That is an indication of charge acceptance.
- In a heavily sulfated battery, to start with, the terminal voltage will be very high (36 V for a 24 V battery). As the charging progresses and the lead sulphate amount slowly come down into the electrolyte solution, the voltage comes down to about 24 V and then slowly picks up. Similarly, specific gravity readings will also begin to rise.
- Now, the ampere value can be increased to 10 per cent of the capacity of the battery.
- Care should be taken not to allow the temperature to exceed 50 to 55° If it exceeds, reduce the current or altogether stop the charging for 4 to 6 hours, or until the temperature subsides to 40°C.
- When there is no further increase in the specific gravity and terminal voltage readings, charging can be terminated.
- After 12 to 24 hours, measure the specific gravity and terminal voltage. If these are normal for the particular battery, it means the battery has been revived.
- If not, discharge the battery until 1.8 volts per cell and recharge it to 130 per cent of the output.
- Again, after about 12 to 24 hours rest period, measure the specific gravity and terminal voltage.
- If they are satisfactory, the battery has been resuscitated.
Should I take up the task of forklift battery reconditioning?
It is strongly advised not to do this. It causes environmental damage at the user site, which will not be prepared for environmentally sound practices. This is best left to be done at the battery manufacturers. They will have adequate facilities to do this in an environmentally safe facility to take care of any accidental spills. This subject has been discussed more for making aware of the possibility of reviving the dead battery. Please consult the forklift battery manufacturers for more information on this.




