Solid state battery introduction
In a battery, positive ions move between the negative and positive electrodes via an ion conductor and deliver electrons to generate an electric current. In conventional batteries example lithium-ion batteries, the ionic conductor is a highly combustible liquid organic compound which is a significant disadvantage. Various research and development processes adopted synthesized a variety of compounds to find high-performance solid conductors to replace liquid conductors. Researchers have discovered a solid-state ion conductor that surpassed the performance of a conventional lithium-ion conductor. ex: LGPS sulfide solid electrolyte (LGPS: lithium, germanium, phosphorus, sulfur)
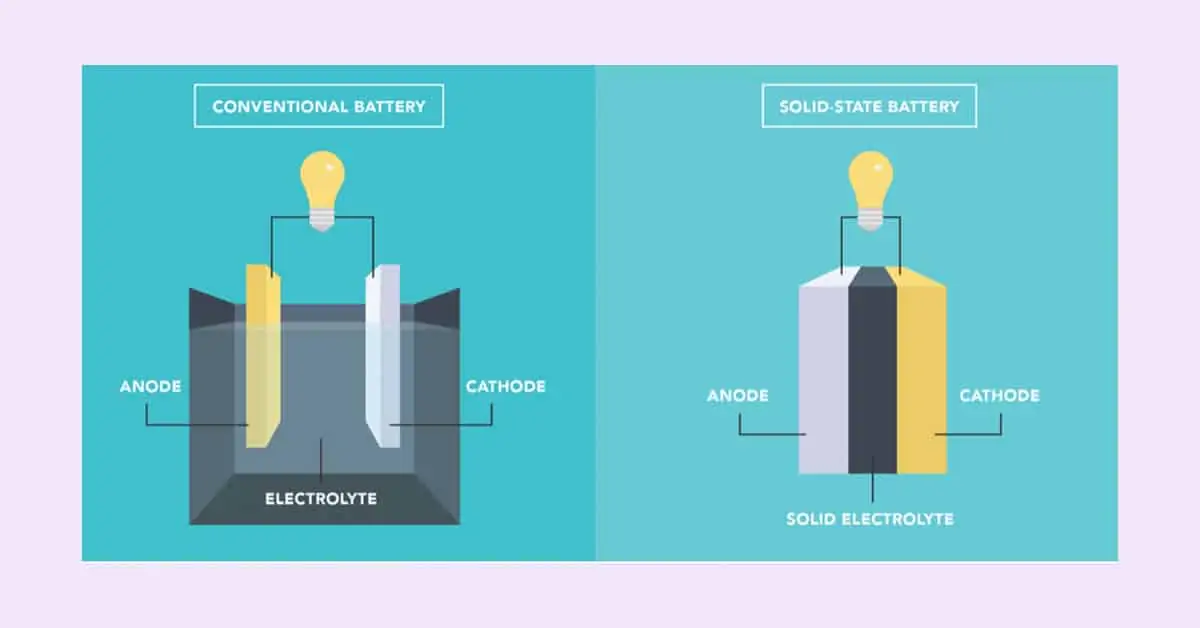
What is a solid state battery? It is the technology approach that has a higher potential for greater safety, higher energy density, and cost-effectiveness. Solid-state batteries are the future for battery technology in consumer electronics and electric vehicles. Cathode, anode, separator, and electrolyte make up a lithium-ion battery. The liquid electrolyte solution is used in liquid state batteries (lithium-ion batteries), which are applied in smartphones, power tools, and electric vehicles. A solid-state battery, on the other hand, uses a solid electrolyte rather than a liquid electrolyte as used in conventional batteries.
The electrolyte in a battery is a conductive chemical mixture that allows current to pass between the anode and cathode. Separators avoid a short circuit. Solid-state batteries are electrochemical cells with an anode, cathode, and electrolyte, much like any other battery. The electrodes and electrolytes are solid, unlike lead-acid batteries.
The commercially available Li-ion battery has a separator that holds the cathode and anode separated by a liquid electrolyte solution. Solid-state batteries, on the other hand, use solid electrolytes rather than liquid electrolyte solution, and the solid electrolyte also acts as a separator. These batteries are essential and strongly needed to increase the capacity of solid state EV battery. They are inflammable, and the chances to explode are negligible. An example of a solid-state battery is lithium phosphate glass. The energy density is high in these batteries.
The energy capacity of a solid-state battery is greater than that of a Li-ion battery with a liquid electrolyte solution. Since there is no chance of explosion or fire, there is no need for safety components, which saves space. The batteries can pack in twice as much energy compared to lithium-ion batteries resulting in increasing their power. Since only a few batteries are needed, a solid-state battery can increase energy density per unit area.
Solid state battery technology
SSB mainly focus on the following characteristics:
Higher energy density:
- Lower cost: use of cheaper materials and cost-effective processes and due to high energy density.
- Higher safety: tolerance to overcharging, tolerance to deep charge
- Lower dependence of scarce materials: less geological dependence, substitution for materials like lithium, cobalt.
- Lower environmental impact: no toxic materials, no heavy metals, no dangerous chemicals, environmentally friendly production, materials easy to dispose of or recycle.
- Others: deep discharge capability, fast charge, or discharge capability.
The solid-state electrolyte is the key component in solid-state batteries. There are three main types of solid-state electrolyte materials.
Inorganic materials: inorganic crystalline materials, inorganic amorphous materials. Since inorganic electrolytes have high elastic moduli, strong thermal/chemical stability, a large electrochemical window, high ionic conductivity, and low electronic conductivity, these electrolytes are better suited for rigid battery designs that can work in harsh environments.
Solid polymers: ex: polyethylene oxide. Although polymer electrolytes possess ionic conductivity lower than that of inorganic solid electrolytes, they can provide a variety of geometries, high flexibility, and require low-cost and simplified production processing. When integrating battery cells, solid polymer electrolyte can easily create an effective electrode-electrolyte link, which could increase the electrochemical stability and cycle life of the batteries. A liquid electrolyte is used in traditional lithium-ion batteries, and it typically makes good contact with the electrodes.
The electrodes absorb the liquid like a sponge thanks to their textured surfaces, resulting in a large contact area. In theory, two solids cannot be connected seamlessly. As a result, the resistance between the electrodes and the electrolyte is high. The solid electrolyte acts as a stable carrier medium for the phosphate electrodes, which are screen printed on both sides. The new solid-state battery, unlike traditional lithium-ion batteries, is entirely free of poisonous or hazardous substances.
Inorganic and polymer materials are combined to fetch the advantage of both types of materials. These electrolytes show high ionic conductivity and are relatively flexible.
The mechanical, electrical, and chemical properties of the solid electrolyte, as well as their interfaces/interphases with the anode and cathode electrodes, have a significant impact on the efficiency of solid-state battery.
The basic issues in solid-state batteries, with an emphasis on three essential phenomena:
(i) the principles of producing advanced ionic conductors,
(ii) structural progression at chemically unstable electrolyte-electrode interfaces, and
(iii) the implications of processing solid-state batteries, including electrode and electrolyte architecture. Solid-state electrolytes (SSEs) can not only resolve safety issues but also allow for the use of a metal anode and high-voltage operation.
The solid-state battery (SSB) is one of the best solutions for the next generation of batteries since solid electrolytes have a much higher thermal stability. Furthermore, inorganic solid electrolytes may function in extreme temperatures, such as those ranging from 50 to 200°C or even higher, where organic electrolytes fail due to freezing, boiling, or decomposition.
To achieve the expected electrochemical output when using all-solid-state electrolytes, four unique features are considered. These characteristics include:
( i )high ionic conductivity (+Li > 104 S/cm);
(ii) adequate mechanical strength and few structural defects to prevent lithium dendrite penetration;
(iii) low-cost raw materials and easy preparation processes; and
(iv) the low activation energy for lithium-ion diffusion.
Solid state battery advantages
- Simple structure: solid electrolytes act as a separator preventing contact of the anode and cathode which results in higher energy density and avoiding the cost of the separator.
- High voltage: decomposition of solid electrolytes is high which in turn leads to high energy density.
- Non-flammable solid electrolyte.
- Electrolyte is flame retardant.
- No risk of liquid electrolyte leak.
- Can be used at higher operating temperatures leading to a larger operating temperature range.
- Possibility of cell stacking in one package.
- Simple cell structure and simple manufacturing costs make solid-state battery cost-effective.
- Solid-state battery charge 6 times faster than liquid state batteries.
- Lifespan of a solid-state battery can last up to 10 years.

Solid state battery disadvantages
- Dendrites are the most serious issue with solid state batterry, aside from cost. Dendrite is a lithium metal crystallization that begins at the anode and can spread across the battery. This happens as ions in the solid electrolyte join with electrons to create a sheet of solid lithium metal because of high current charging and discharging.
- These batteries are not extensively used in consumer electronics and electric vehicles as they are very expensive. solid state batteries exhibit Slower kinetics due to:
- Low ionic conductivity
- High interfacial resistance
- Poor interfacial contact
How do solid state battery work?
As previously mentioned, solid-state batteries use a solid electrolyte membrane with solid positive and negative electrode materials. During charge or discharge, ions migrate into an ion-conductive solid matrix rather than an ionic salt dissolved in a solution, causing the charge or discharge reactions to take place. Redox reactions are used to store and distribute energy in solid-state batteries. The anode undergoes oxidation, while the cathode undergoes reduction, and the battery can use this phenomenon to store (charge) and release (discharge) energy as required.
While discharging energy, ions cause a chemical reaction between the battery’s materials called ‘Redox,’ in which oxidation occurs at the anode to create compounds with free electrons, which supply electric energy, and reduction occurs at the cathode to create compounds that gain electrons and therefore preserve power. The mechanism is reversed when a battery is charged. Positively charged ions pass through the electrolyte from the negative electrode (anode) to the positive electrode (cathode) when discharging solid-state batteries (cathode). This causes a positive charge to develop in the cathode and absorbs electrons from the anode.
However, since electrons cannot pass through the electrolyte, they must travel via a circuit, providing power to whatever it is attached to, such as an electric motor. In the process of charging, ions migrate to the anode, accumulating a charge that absorbs electrons from the cathode through a circuit. The battery is assumed completely charged when no more ions can flow to the negative electrode. Solid-state batteries require different additives and binders within their layers to retain high conductivity during cycling. The materials must also be held under pressure to maintain contact during charging and discharging periods. The normal expansion and contraction of materials during charging and discharging cycles add to the difficulties of maintaining a firm touch.
The cycle life and output of the cell may be harmed if the expansion and contraction weaken the bond over time. Solid-state batteries also simplify the pack level, where individual cells are attached, compared to Li-ion batteries. Solid-state batteries don’t need much in the way of thermal control because their efficiency improves as the temperature rises.
The overall charge and discharge rates, as well as the ionic conductivity of solid electrolytes, increase as the temperature rises. As a result, a solid-state cell’s ultimate operating temperature is only constrained by the melting point of lithium, which is 180°C. Furthermore, the absence of a flammable Li-ion liquid electrolyte eliminates design concerns about the catastrophic cell or pack failure. Lithium metal-based solid-state batteries should be used as Li-ion battery alternatives because the graphite anode used in typical Li-ion batteries has a low potential compared to lithium (0.20 V), offering greater volumetric energy density with equivalent voltage and performance.
Are solid state batteries available?
Pacemakers, RFID, and portable devices use solid-state batteries. Some of these batteries are being utilized in space applications. Commercialization approaches for solid-state batteries in EV/HEV automobile market. To bring solid-state batteries is not just about developing a suitable solid-state electrolyte but considering features like:
- Securing material supply and sales.
- Cell and pack manufacturing equipment and development.
Despite years of development, many players have not succeeded to bring solid-state batteries to the market. At room temperature, the ionic electrolytes typically are several orders of magnitude lower than that of liquid electrolytes. This is one of the main hurdles for the commercialization of solid-state batteries. While the concept of solid-state batteries has been there for decades, progress is only now being made, thanks to investments from electronics firms, car manufacturers, and general industrial providers.
Why are solid-state batteries better?
Solid-state batteries offer several benefits over their liquid-filled counterparts, including longer battery life, quicker charging times, and a smoother experience. Instead of suspending the electrodes in a liquid electrolyte, solid-state batteries compress the anode, cathode, and electrolyte into three flat layers. As a result, they can be made smaller—or at least accentuate still carrying the same amount of energy as a larger solvent battery.
So, when a phone or laptop has a lithium-ion or lithium-polymer battery with a solid-state battery of the same capacity, it will last much longer. A system is created that carries the same amount of charge but is much smaller and thinner. Solid state batteries, when used to power current devices or even electric vehicles, the batteries could recharge much faster because ions could travel from the cathode to the anode much faster. A solid-state battery could exhibit various rechargeable batteries in terms of capacity by 500 percent or more and charge in a tenth of the time. Solid-state batteries are less harmful to the environment.
Solid-state thin-film batteries are less environmentally hazardous than conventional batteries. Since solid-state batteries have higher performance and energy density, they don’t need the cooling and control components that lithium-ion batteries do, resulting in a smaller overall size, more device freedom, and lower weight.
Since solid-state batteries are resistant to electrode corrosion caused by chemicals in the liquid electrolyte or the build-up of solid layers in the electrolyte that shortens battery life, solid-state batteries can handle more discharge and charge cycles than lithium-ion batteries. Solid-state batteries may be recharged up to seven times more than lithium-ion batteries, allowing them ten years instead of the few years that lithium-ion batteries are meant to last. Educational institutions, battery manufacturers, and material experts are all investigating solid-state batteries could be transformed into next-generation power sources for widespread use.
Do solid state batteries charge faster?
Some solid electrolytes have ionic conductivities greater than 5 mS/cm and are single-ion conductors. At high current, this prevents polarisation resistance from building up in the solid electrolyte. Consequently, fast charging is potentially feasible. Solid-state batteries use a solid material, typically a polymer or ceramic compound, to replace the flammable liquid electrolyte in lithium-ion batteries. Lithium-metal anodes have been introduced as a substitute for traditional graphite or silicon anodes. This effort to develop solid-state lithium-metal batteries has the potential to double energy density while reducing charging time significantly.
How is a solid state battery made?
In solid state battery, there are only two main layers, cathode a positive electrode with an electrical contact with a solid-state ceramic separator which replaces the polymer separator, which replaces the porous polymer separator that is found in conventional lithium-ion batteries. All-solid-state electrolytes rely on superionic conductors and optimized interfaces.
The challenge to develop a good solid-state electrolyte is to overcome the interface, triple-phase boundaries where positive electrodes, ions, and electrons have to be simultaneously transported, which requires a very intermittent phase. Achieving simultaneous electron and ion transport at the interface and control of interfaces is a challenge.
The issues of chemical stability with positive electrolyte and oxidative stability of electrolyte in the presence of carbon and the issues of metal ion dendrites require the incorporation of protected interfaces. Solid electrolytes have a lower ion conductivity than liquid electrolytes by order of magnitude. It is also essential to optimize the resistance at the electrolyte-electrode interface.
Challenges for a solid electrolyte for batteries:
Thick composite positive electrodes with high active mass: high ionic conductivity of solid-state electrolytes. Stable interface with oxide and low redox activity with electronically conducting additives.
Thin low mass solid electrolyte membranes should possess good mechanical properties, ductility, and dynamic pressure control.
All SSB electrolytes offer an unprecedented combination of high ionic conductivity and excellent stability between anode and cathode. The compatibility of the anode is the key in this because it provides a primary benefit at the cell level over other conventional lithium-ion batteries.
There are three major solid electrolytes:
Polymer electrolyte: the advantage of polymer electrolyte is cell processability. The drawbacks are relatively poor stability against the metal and relatively poor conductivity at especially lower temperatures.
Lower ionic conductivity=lower transport of ions = less power.
Oxide electrolyte: they possess ideal mechanical properties, are very rigid, and are chemically stable against metal anode. The major drawbacks include low-grade capabilities in terms of how quickly they can be corroded using an oxide electrolyte and challenging to process because they require very high temperatures. They possess higher thermal stability, negligible cell processability, sensitive to moisture, and are moderate in terms of conductivity. Oxide-based electrolytes are normally chemically stable and can be used with high-energy cathode materials.
The ion conductivity is, however, lower than that of sulfide-based electrolytes.
Materials with a perovskite (LLTO: Lithium Lanthanum Titanium Oxide)
Garnet structure (LLZO, lithium lanthanum zirconium oxide), as well as NASICON (LAGP: Lithium Aluminum Germanium Phosphate), are impressive among oxide-based electrolytes.
Sulfide electrolyte: they possess mechanical properties between that polymer and an oxide. They are more conductive of any electrolyte class. All the record-breaking electrolytes come from sulfide class materials. They possess higher conductivity, higher cell processability, and higher thermal ability but are sensitive to moisture. Ionic conductivity is normally higher in sulfide-based electrolytes, but they are more chemically unstable.
At room temperature, the amorphous lithium tin phosphorus sulfide (LSPS) has a very high ion conductivity. Incompatibilities with lithium metal, on the other hand, are a concern.
A material that determines electrolytes is known as an additive. An additive is a small amount of a material that forms a protective coating on the cathode and anode surfaces. It prevents battery degradation by facilitating the smooth passage of lithium ions between the cathode and the anode.
Cathode and anode additives are the two types of additives. Cathode additives prevent battery aging by stabilizing the cathode structure and protecting the surface, eliminating overheating and overcharging. Anode additives dissolve faster than a solvent, forming a strong film in the anode that extends its life, prevents overheating, and keeps the battery charged. additives play an important role in the overall system by extending its lifetime, improving high-temperature problems, and lowering resistance.
A separator is a thin insulating membrane with about four features that hold the cathode and anode separated. Second, as the name suggests, separators protect the cathode and anode from making contact within the battery.
Second, separators have submicron-sized pores that are invisible to the naked eye, and the pores serve as channels for lithium ions to pass between the cathode and the anode. Since separators have good mechanical stability, the tensile property keeps by-products and foreign substances out, ensuring safety. Electrochemically stable and high-insulation materials can be used as separators. Separators are supposed to avoid cathode and anode interaction, and it leads to major issues if they interfere with lithium ions or other ions within a battery. Separators should be able to ensure protection by shutting down pores and preventing the movement of ions if the temperature of a battery exceeds a certain limit.
Finally, SSB separators should be small enough to allow more active materials to be incorporated into a battery, increasing energy density. To avoid damage and ensure protection, they should also have high mechanical power.
Solid electrolyte requirements
Solid electrolytes with a specific combination of properties will be needed for the commercialization of solid-state batteries. To be a suitable liquid electrolyte substitute, solid electrolytes must have lithium ionic conductivities greater than 0.1 mS/cm. Either the electrolyte must be chemically stable to lithium reduction, or a passivating reaction layer must be formed. To keep internal cell resistance down, the electrolyte needs to form low-resistance interfaces.
At the alkali metal interface, where atmosphere-reacted substrate layers, reduced oxides, and inhomogeneous wetting can all lead to substantial interface resistance, creating low-resistance interfaces adds complexity., the electrolyte must have sufficient strength and fracture toughness to avoid the dissemination of lithium filaments through the electrolyte. At both the anode and cathode potentials, the electrolyte must be stable.
Solid electrolyte forms
Since polymer solid electrolytes have low ionic conductivities, they are usually used at higher temperatures (60°C–80°C) to gain the benefit of higher ionic transport. Though polymers are simple to work with, their mechanical properties are inadequate to keep the lithium metal anode stable.
As a result, inorganic solid electrolytes have received the most recognition. The conductivities of solid sulfide electrolytes are among the strongest of all solid electrolytes.
Although there are many chemistries, the Li2 S-P2 S5 system is the most used. In the Li2 S-P2 S5 framework, the electrolytes can be glassy, crystalline, or partly crystalline. Li2 S-P2 S5 electrolytes that are not doped have low electrochemical stability with lithium, while doped versions have improved stability. At room temperature or below 400°C, the ductile nature of sulfide electrolytes allows them to compress into compacts with good electrochemical bridging between particles. As a result, sulfide electrolytes are the easiest inorganic solid electrolytes to process.
However, reactivity with water vapour in the air can be a problem with certain sulfide electrolyte compositions, releasing H2 S and degrading the electrolyte. As a result, they’re normally processed in argon or low-humidity dry room environments.
Oxide-based solid electrolytes are a second form of an inorganic solid electrolyte. There are a few different forms, but garnet Li7 La3 Zr2 O12 is the most common. At room temperature, solid oxide electrolytes have strong ionic conductivities, the widest electrochemical range, and the maximum chemical stability against lithium. Furthermore, oxide materials have the highest elastic moduli and fracture toughness of any solid electrolyte, making them ideal for the physical stability of the lithium metal anode and long-term cell lifetimes. Despite having the best mixture of electrochemical properties, dense electrolytes with high ionic conductivity need sintering temperatures of 1,000°C – 1,300°C.
Resistance to dendrite or lithium filament development in solid electrolytes tends to be linked to current density or total cell current divided by electrolyte cross-sectional region. As a result, the cell can fail at a critical current density (CCD) when the lithium metal penetrates the cell. Stable charging is possible at current densities under this critical value. Constant current plating of lithium in asymmetric cells with lithium electrodes on either side of a solid electrolyte is a standard CCD examination.
In the preparation of LGPS sulfide solid electrolyte, the material’s structure was analyzed with neutron beams. Researchers were able to observe the linear motion of ions inside the solid conductor’s molecular structure. A tunnel is observed within its 3D structure. The motion of lithium ions was observed within this tunnel. With this hint, researchers were able to make improvements to the material’s ionic conductivity and stability, developing two new materials by adding a small amount of chlorine to LGPS. These materials exhibited the world’s highest ionic conductor performance.
These materials were analyzed with a materials design diffractometer. The results showed an innovative structure that allowed ions to move in three dimensions rather than just one. This is what made the highest performance of the materials possible. Among a wide range of new generation cells that have been developed, these materials became strong electrolytes for use in all SSB.
These SSB have slightly higher energy density and higher power output when compared to lithium-ion batteries. Thus, merits of all SSB are designed to have compact, high-capacity batteries that can be recharged in a short period. As a product of dedicated research and atomic analysis level of electrolyte materials, these new all SSB could lead to a new generation of batteries.
In lithium ion solid-state battery, as the battery charges, the lithium leaves the traveling through the atomic lattice of a nonporous solid-state ceramic separator. Once the lithium is truly separated it deposits between the separator and the electrical contact forming an anode of pure metallic lithium. For lithium metal anode allows the energy of a solid-state battery to be stored in a smaller energy volume enabling a higher energy density as compared to conventional lithium-ion batteries. Solid-state lithium metal batteries allow a greater range from the higher energy density of fifteen-minute fast charge and safer operation by eliminating the organic polymer separator.
High energy lithium-ion battery:
- Use of high-energy materials
- Nickel-rich NMC or NCA cathodes, when combined with a silicon composite anode, offer higher Gravimetric and volumetric energy densities.
- Cost reductions are expected for manufacturing purposes.
- Only minor changes to the production process are needed.
Solid state Lithium battery
Lithium-sulfur battery:
- Upon discharge, lithium reacts with sulfur at the cathode to form lithium sulfides.
- Sulfur, as a widespread and cost-effective material, offers low-cost battery cells.
- Uncertainty regarding cycle and life, and a high-temperature sensitivity, continue to thwart market segment growth.
- Currently, the volumetric energy density tends to be too poor for automobile use.

Lithium-air battery:
- Lithium is oxidized with oxygen at the cathode side upon discharge, resulting in lithium peroxide and lithium oxide.
- High energy density and the use of ambient air are technically feasible.
- Huge obstacles in cycle stability, which makes a car application in the next decade seem improbable.
Functionality and design of all SSB
- An ion-permeable solid electrolyte acts as a separator and provides both spatial and electrical separation between the cathode and anode of an all SSB
- There are a variety of cell designs to choose from. A thin-film cell is depicted in the diagram above. A composite cathode may be used to create thicker layers.
- Lithium ions pass from the anode via the solid electrolyte to the cathode when an all SSB is discharged. A power flows at the external load at the same time.
- The resistance at the anode-electrolyte interface is a critical factor in the battery cell’s efficiency. An external sheet, such as a rubber or an aluminum alloy, may be used to mitigate this.
- Bipolar stacking is possible due to the strong electrolyte. Consequently, the elementary cells are connected serially.
What are solid state batteries made of?
Solid-state battery materials:
Anode:
Because of their theoretical potential for achieving the maximum energy density, lithium metal anodes are considered ideal. The strong electrolyte, on the other hand, must prevent metallic lithium from forming dendrites. Furthermore, since lithium forms a passive layer with atmospheric oxygen, handling under an inert atmosphere is required.
Silicon as an anode material offers very high energy densities, but it goes through a lot of volume shifts when it’s mixed with lithium.
Cathode:
The metal oxide is used as the cathode. Since there are much fewer materials that have been specifically designed for the all SSB, existing cathode materials are used in most cases.
In principle, a wide variety of proven cathode materials can be used, based on the electrolyte, varying from inexpensive and safe materials like lithium iron phosphate (LFP) to lithium nickel manganese cobalt oxide (NMC). Only lithium cobalt oxide (LCO) as a cathode material and LLZO as an electrolyte demonstrate adequate stability and efficiency in practice.
Process of manufacturing of all solid state battery
- Electrode and electrolyte processing, cell assembly, and cell finishing are the three major steps in the production of an all SSB.
- There is no universally true process chain; alternatively, a wide number of possible process chains may be used. These vary from the lithium-ion battery production process in several ways.
- This method compares and contrasts two different process choices, primarily in terms of electrode and electrolyte output.
Electrode and electrolyte
Production —- cell assembly——cell finishing
Process A
Process B
The synthesis of trench cells with inorganic solid electrolytes is the subject of both process options. For all-solid-state batteries, the pouch cell format tends to be the most appropriate.
A prismatic or round cell:
Due to the solid components of an all-solid-state battery, windings face major challenges. Ceramic layers that are brittle can develop cracks. Furthermore, the problem of proper layer adhesion has yet to be resolved.
Pouch cell:
All-solid-state batteries benefit from stacking because the flat layers are not deformed. Furthermore, the layer compound is produced during electrode and electrolyte processing, leaving only the basic cells to be stacked later.
A dry room is needed for the manufacturing process due to the materials’ reactivity to the atmosphere. When working with metallic lithium, an inert gas, such as argon, is recommended.
A comprehensive evaluation of the applicability of skill acquired in the development of lithium-ion battery cells is performed for each process stage.
Electrode and electrolyte production through process A:
- The compound of cathode, electrolyte, and anode is formed in electrode and electrolyte processing.
- An elementary cell exists after electrode and electrolyte development.
- The key feature of the first process chain, Process Chain A, is a continuous extrusion process in which the layers are formed and then laminated.
- This process chain is particularly well suited to sulfide-based all-solid-state materials.
Cathode and electrolyte production(compounding):
- The production method
- Two different compounding industries utilize cathode and electrolyte melts.
- The material components are fed into a twin-screw extruder’s heated barrel and can be provided as granulate or powder.
- The extruder’s rotational motions carry energy into the material components. As a consequence, the melt is homogeneous.
- Electrolyte particles, which lower the resistance between the cathode and the electrolyte, as well as binders and additives, are mixed in with the cathode active content.
- Electrolyte molecules and polymer binders are two material elements of the electrolyte.
Parameters and conditions for the process:
- Quantity of individual materials to be supplied
- Temperature and pressure in the cylinder
- The extruder’s rate and pressure
- Shear power
Quality features:
- The melt’s homogeneity
- The melt’s viscosity
- Amalgamate scale and quantity
Technology alternatives:
- High-performance mixing plant
Cathode and electrolyte production (co-extrusion):
- The production method
- In an appropriate die, the cathode and electrolyte melts are co-extruded. This results in a cathode and electrolyte layer combination.
- Separate channels feed the cathode, and the electrolyte melts through the extrusion die.
- The melts travel through the channels to the extrusion die’s outlet. The melts are extruded onto a current conductor using a slot die.
Process parameter and requirements:
- Adjustment of the layer thickness
- Melt feed rate
- Temperature
- Pressure
- Roll speed
- Pressing pressure of the calender roll
Quality features:
- Coating thickness
- Layer width
- Adhesion between layers
Technology alternatives:
- Screen printing
Foil casting
Anode production (extrusion and calendering):
- An all SSB anode can be made of metallic lithium foil. Extrusion with subsequent calendaring can be used to make this lithium film.
- Liquid lithium is poured into the cylinder of a piston extruder for this purpose. The lithium is then squeezed into a nozzle by a piston.
- Calendaring after extrusion ensures homogeneity and optical film thickness. The film is rolled under tension by two rollers with the application of a lubricant for this reason.
- The rollers must be able to work with lithium’s adhesiveness. Polymer-coated rollers, such as those made of polyacetal, will accomplish this.
Process parameters and requirements:
- Extrusion speed
- Temperature
- Nozzle geometry
- Pressing pressure of the calendar rolls
- Supply speed of the lubricant
- Roll speed
Quality features:
- Film thickness
- Foil width
- Homogeneity of the lithium foil
Technology alternatives:
- Atomic layer deposition
PVD process
Layer compound production (laminating):
- Lithium foil is laminated onto the cathode-electrolyte composite after it has been processed. The two layers are placed together using rollers for this task.
- Two rollers are used to force the two layers together in the next stage. To obtain greater adhesion forces, these are heated. Polymers penetrate from one layer to the next during heating and pressing, creating the link between anode and electrolyte.
- The words “dry” and “wet” lamination can be differentiated. Until lamination, wet lamination moistens the contact surfaces with a solvent. This facilitates low-temperature and low-pressure lamination.
Parameters and requirements of the process:
- Feeding speed of layers
- Roll speed
- Pressure
- Optional heating of the layers
Quality features:
- Adhesion between layers
- Desired composite thickness
- The geometry of the composite
Technology alternatives:
- Pressing and subsequent sintering
Electrode and electrolyte production process B
- The physical vapor deposition (PVD) process, in which the individual layers are added one after the other, is the key feature of procedure chain B shown below.
- This current process, which shows the manufacturing steps of a thin-film battery, is especially suitable for oxide-based all-solid-state batteries.
Material preparation (Grinding and Mixing):
- Methodology of manufacture
- A ball mill is used to isolate the cathode powder from the electrolyte powder.
- The raw materials are placed into a cylindrical grinding drum for this task. Balls are used as grinding media in this grinding drum.
- The rotational motions of the cylinder combine the starting materials. Furthermore, the rotational movement ensures that the grinding media and the starting material shift relative to each other when the latter is ground.
- After that, the powder is calcined to achieve the desired powder properties.
Requirements and parameters of the process:
- Ball material
- Speed
- Grinding time
- Cylinder material
- Quantity of starting material
Quality features:
- Average powder particle size
- Homogeneity of the powder (degree of mixing)
Technology alternatives:
- Sol-gel process
Layer compound production (high-frequency sputtering):
Manufacturing process:
- High-frequency sputtering is used to create the cathode and electrolyte layers from cathode and electrolyte powders. The goal for the sputtering process is first made from powders using a die or hot-pressing system.
- The present collector also acts as the process’s substrate. The cathode layer is deposited in the first phase. An electrolyte layer is then placed on top of the cathode layer.
- Ions are aimed at the target of the sputtering operation. Atoms are knocked out of the target in this step, which then reaches the gas phase and advances to the substrate. The layer is therefore developed atom by atom on the substrate’s surface.
- A vacuum chamber is used for high-frequency sputtering.
Requirements and parameters of the process:
- Temperature
- Deposition time
- Process pressure
- Ambient atmosphere
- Process power/power density
- Target diameter & target distance
Quality features:
- The layer thickness of the current collector
- The layer thickness of the cathode and the electrolyte
Technology alternatives:
- Chemical vapor deposition
Layer compound deposition (sintering)
Manufacturing process:
- The cathode and electrolyte layers are compressed during sintering. By enhancing the bond between the two layers, the resistance at the interface electrolyte–electrode can be reduced.
- A sintering furnace is used to sinter the cathode–electrolyte compound. The substance is heated to just below its melting point.
- The resultant porosity of the materials can be modified based on the process parameters chosen.
- To avoid reactions with the environment, the sintering process occurs in an inert atmosphere or a vacuum.
- Sintering is especially critical for oxide-based solid electrolytes to achieve lower enough interfacial tolerance.
Layer compound production (Thermal evaporation):
Process of production:
- The anode can be applied to the cathode –electrolyte compound using thermal evaporation. The anode content is made of metallic lithium.
- Thermal evaporation requires heating metallic lithium to temperatures above boiling point, such as with an electron beam evaporator, so that it can reach the vapor phase. In the vacuum chamber, steam spreads uniformly.
- Condensation forms the coating on the lower-temperature surface of the electrolyte.
- Thermal evaporation happens in a vacuum chamber, comparable to sputtering.